Comparative antioxidant and antibacterial activities of ethanol extract from different parts of Syzygium gratum
DOI:
https://doi.org/10.55674/cs.v18i2.266044Keywords:
Syzygium gratum, antioxidant, antibacterial, Dihydrooroxylin AAbstract
Herbal extracts are widely recognized as valuable sources of bioactive compounds with antioxidant and antimicrobial properties. However, variations in biological activity among different plant parts remain insufficiently explored for many medicinal plants. This study aimed to comparatively evaluate the antioxidant and antimicrobial activities of extracts obtained from four different parts including young leaves (YL), mature leaves (ML), branches (BR), and twigs (TW) of Syzygium gratum. Plant materials were extracted using ethanol with maceration method, and antioxidant activity was determined through total phenolic content (TPC) analysis along with the 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging assay, while antibacterial activity was evaluated against two selected microorganisms, E. coli, S. aureus using disc diffusion method. Among the tested extracts, the extract from young leaves exhibited the strongest antioxidant capacity with 18.01±2.25 mg gallic acid equivalents per gram of dried weight for TPC and 67.05% for DPPH assay. In addition, the extract showed the most antimicrobial effects with 10.8±0.1 and 11.1±0.1 mm for E. coli and S. aureus, respectively. The YL extract was further investigated the chemical constituents by using GC–MS technique, and the result revealed that the four major constituents were Dihydrooroxylin A, neophytadiene, tetrapentacontane, and g-tocopherol. The observed data was likely associated with major GC–MS identification constituents, including Dihydrooroxylin A and γ–tocopherol, which have been previously reported to exhibit antioxidant and antimicrobial effects. In addition, these data highlighted the importance of plant–part selection especially for YL extract in maximizing biological activity and supported the potential application of S. gratum as a natural source of antioxidant and antimicrobial agents.
GRAPHICAL ABSTRACT
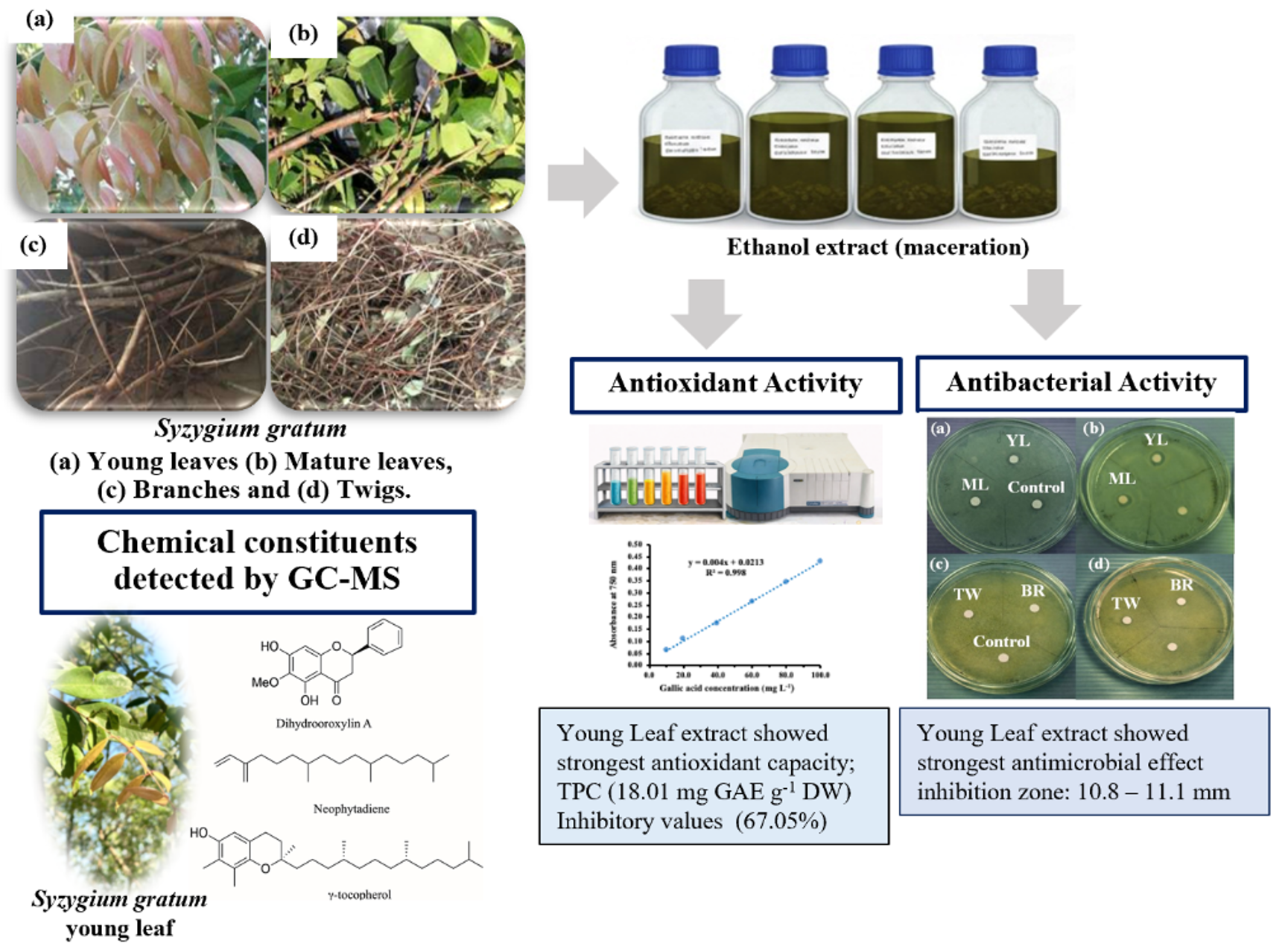
HIGHLIGHTS
- Ethanol extracts obtained from different parts of Syzygium gratum exhibited moderate antioxidant and antibacterial activities.
- Extracts from young leaves showed higher biological activities comparing with other parts, in agreement with their phenolic content and the presence of bioactive constituents detected by GC–MS analysis.
- These results indicate the relevance of plant-part selection for the rational and sustainable utilization of Syzygium gratum in environmentally related chemical applications.
References
Li, S., Jiang, S., Jia, W., Guo, T., Wang, F., Li, J., & Yao, Z. (2024). Natural antimicrobials from plants: Recent advances and future prospects. Food Chemistry, 432, 137231. https://doi.org/10.1016/j.foodchem.2023.137231
Intharuksa, A., Kuljarusnont, S., Sasaki, Y., & Tungmunnithum, D. (2024). Flavonoids and other polyphenols: Bioactive molecules from traditional medicine recipes/medicinal plants and their potential for phytopharmaceutical and medical application. Molecules, 29(23), 5760. https://doi.org/10.3390/molecules29235760
Cowan, M. M. (1999). Plant products as antimicrobial agents. Clinical Microbiology Reviews, 12(4), 564–582. https://doi.org/10.1128/cmr.12.4.564
Dai, J., & Mumper, R. J. (2010). Plant phenolics: extraction, analysis and their antioxidant and anticancer properties. Molecules, 15(10), 7313–7352. https://doi.org/10.3390/molecules15107313
Bakkali, F., Averbeck, S., Averbeck, D., & Idaomar, M. (2008). Biological effects of essential oils–A review. Food and Chemical Toxicology, 46(2), 446–475. https://doi.org/10.1016/j.fct.2007.09.106
Rocchetti, G., Lucini, L., Ahmed, S. R., & Saber, F. R. (2019). In vitro cytotoxic activity of six Syzygium leaf extracts as related to their phenolic profiles: An untargeted UHPLC-QTOF-MS approach. Food Research International, 126, 108715. https://doi.org/10.1016/j.foodres.2019.108715
Chotchoungchatchai, S., Saralamp, P., Jenjittikul, S. Pornsiripongse, S., & Prathanturarug, S. (2012). Medicinal plants used with Thai traditional medicine in modern healthcare services: A case study in Kabchoeng Hospital, Surin Province, Thailand. Journal of Ethnopharmacology, 141, 193–205. https://doi.org/10.1016/j.jep.2012.02.019
Uddin, A. N., Hossain, F., Reza, A. A., Nasrin, M. S., & Alam, A. K. (2022). Traditional uses, pharmacological activities, and phytochemical constituents of the genus Syzygium: A review. Food Science & Nutrition, 10(6), 1789–1819. https://doi.org/10.1002/fsn3.2797
Kukongviriyapan, U., Luangaram, S., Leekhaosoong, K., Kukongviriyapan, V., & Preeprame, S. (2007). Antioxidant and vascular protective activities of Cratoxylum formosum, Syzygium gratum and Limnophila aromatica. Biological and Pharmaceutical Bulletin, 30(4), 661–666. https://doi.org/10.1248/bpb.30.661
Senggunprai, L., Kukongviriyapan, V., Prawan, A., Kukongviriyapan, U. (2010). Consumption of Syzygium gratum promotes the antioxidant defense system in mice. Plant Foods for Human Nutrition, 65(4), 403–409. https://doi.org/10.1007/s11130-010-0200-6
Wongklom, A., Siriwan, K., & Krairam, S. (2023). Effect of drying methods on the water soluble phenolics, flavonoids and antioxidant activities in baby jackfruit leaves (Momordica cochinchinensis (Lour.) Spreng.) powder. Creative Science, 15(1), 240449. https://doi.org/10.55674/cs.v15i1.240449
Tassana-iem, T., Sangkla, K., Srina, W., Charoenrat,Y., Sittiwong, P., & Sawatmoon, S. (2026). Total phenolic content and phytochemicals of broken landrace rice and their utilization in food products. Creative Science, 18(1), 263023. https://doi.org/10.55674/cs.v18i1.263023
Khunchaelae, J., & Surapat, V. (2023). Study of free radical scavenging, total phenolic contents, and tyrosinase inhibition activity of crude extract from Moringa oleifera Lam. Creative Science, 15(1), 241942. https://doi.org/10.55674/cs.v15i1.241942
Martins, G. R., Monteiro, A. F., do Amaral, F. R. L., & da Silva, A. S. A. (2021). A validated Folin–Ciocalteu method for total phenolics quantification of condensed tannin-rich açaí (Euterpe oleracea Mart.) seeds extract. Journal of Food Science and Technology, 58(12), 4693–4702. https://doi.org/10.1007/s13197-020-04959-5
Lawag, I. L., Nolden, E. S., Schaper, A. A. M., Lim, L. Y., & Locher, C. (2023). A modified Folin–Ciocalteu assay for the determination of total phenolics content in honey. Applied Sciences, 13(3), 2135.https://doi.org/10.3390/app13042135
Sihanat, A., Rittaisong, N., Chaichamnong, N., Prommee, N., Watcharaput, N., Noppakao, P., & Yamprasert, R. (2023). In vitro antioxidants activities of extract of Tri-Kasorn-Mas remedy and its plant ingredients. Creative Science, 15(2), 249899. https://doi.org/10.55674/cs.v15i2.249899
Phinetsathian, K., Arbsuwan, N., Somrug, K., Chaiyapet, N., Taikham, P., Chailoed, S., & Jirawattanasomkul, N. (2025). Growth and effective inhibition of pathogens of Cordyceps militaris cultured on Sakon Nakhon local rice. Creative Science, 17(2), 261051.
https://doi.org/10.55674/cs.v17i2.261051
Ayoola, G. A., Coker, H. A., Adesegun, S. A., Adepoju-Bello, A. A., Obaweya, K., Ezennia, E. C., & Atangbayila, T. O. (2008). Phytochemical screening and antioxidant activities of some selected medicinal plants used for malaria therapy in Southwestern Nigeria. Tropical Journal of Pharmaceutical Research, 7(3), 1019–1024. https://doi.org/10.4314/tjpr.v7i3.14686
Tiranakwit, T., Puangpun, W., Tamprasit, K., Wichai, N., Siriamornpun, S., Srisongkram, T., & Weerapreeyakul, N. (2023). Phytochemical screening on phenolic, flavonoid contents, and antioxidant activities of six indigenous plants used in traditional Thai medicine. International Journal of Molecular Sciences, 24(17), 13425. https://doi.org/10.3390/ijms241713425
Kumutanat, W., Hongthong, S., Thanasansurapong, S., Kongkum, N., & Chumnanvej, N. (2023). GC-MS and bioassay-guided isolation of xanthones from Mammea siamensis. Indonesian Journal of Chemistry, 23(3), 716–726. https://doi.org/10.22146/ijc.79987
AOAC International. (2015). Standard method performance requirements (SMPRs) for estimation of total phenolic content using the Folin–Ciocalteu assay (AOAC SMPR 2015.009). Official Methods of Analysis of AOAC International.
Sriket, P. (2012). Chemical components and antioxidant activities of Thai local vegetables. International Food Research Journal, 19(1), 18–26.
Pakdeechote, P., Meephat, S., Sakonsinsiri, C., Phetcharaburanin, J., Bunupha, S., & Maneesai, P. (2020). Syzygium gratum extract alleviates vascular alterations in hypertensive rats. Medicina,56(9), 509. https://doi.org/10.3390/medicina56090509
Jia, D., Arbab, S., Ullah, H., Alzahrani, K. J., Alzahrani, F. M., Alsharif, K. F., Zhang, J., & Li, K. (2025). Antibacterial activity of traditional medicinal plants: Combating antibiotics resistance in animal wound infections. Veterinary Medicine and Science, 11(3), e70361. https://doi.org/10.1002/vms3.70361
Álvarez-Martínez, F. J., Barrajon-Catalán, E., Herranz-López, M., & Micol, V. (2021). Antibacterial plant compounds, extracts and essential oils: An updated review on their effects and putative mechanisms of action. Phytomedicine, 90, 153626.
https://doi.org/10.1016/j.phymed.2021.153626
Górniak, I., Bartoszewski, R., & Króliczewski, J. (2019). Comprehensive review of antimicrobial activities of plant flavonoids. Phytochemistry Reviews, 18, 241–272. https://doi.org/10.1007/s11101-018-9591-z
Shamsudin, N. F., Ahmed, Q. U., Mahmood, S., Shah, S. A. A., Khatib, A., Mukhtar, S., Alsharif, M. A., Parveen, H., & Zakaria, Z. A. (2022). Antibacterial effects of flavonoids and their structure–activity relationship study: A comparative interpretation. Molecules, 27(4), 1149. https://doi.org/10.3390/molecules27041149
Al-Zaben, M., Zaban, M. A., Naghmouchi, S., Asaloom, A. N., Al-Sugiran, N., & Alrokban, A. (2023). Comparison of phytochemical composition, antibacterial, and antifungal activities of extracts from three organs of Pistacia lentiscus from Saudi Arabia. Molecules, 28(13), 5156. https://doi.org/10.3390/molecules28135156
Arumugam, R., Elanchezhian, B., Samudurai, J., & Amirthaganesan, K. (2022). Comparative antioxidant, antibacterial and phytochemical analysis of roots, stems, leaves and seeds from Cleome rutidosperma DC. Natural Resources for Human Health, 2(1), 1–14. https://doi.org/10.53365/nrfhh/2022/1/6
Teffo, T. K., Dukhan, S., Ramalepa, P., & Risenga, I. (2022). Comparative phytochemical analysis, antioxidant and antibacterial activities in the leaves, underground stems and roots of Bulbine abyssinica. Biomedical & Pharmacology Journal, 15(3), 1323–1335. https://doi.org/10.13005/bpj/2469
Tambe, B.D., Pedhekar, P., Harshali, P. (2021). Phytochemical Screening and Antibacterial Activity of Syzygium cumini (L.) (Myrtaceae) Leaves Extracts, Asian Journal of Pharmaceutical Research and Development, 9(5), 50–54. http://dx.doi.org/10.22270/ajprd.v9i51023
Sriket, C., & Kittiphattanabawon, P. (2018). Phytochemicals and antioxidant activities of edible herbs. Current Applied Science and Technology, 18, 10–17. [No DOI available].
Zhang, B. X., Li, J., Gu, H., Li, Q., Zhang, Q., Zhang, T. J., Wang, Y. & Cai, C. K. (2015). Using bioinformatics approach to explore the pharmacological mechanisms of multiple ingredients in Shuang-Huang-Lian. The Scientific World Journal, 2015(1), 291680.
https://doi.org/10.1155/2015/291680
Rajeswaran, S., & Rajan, D. K. (2025). Neophytadiene: Biological activities and drug development prospects. Phytomedicine, 143, 156872.
https://doi.org/10.1016/j.phymed.2025.156872
Micera, M., Botto, A., Geddo, F., Antoniotti, S., Bertea, C. M., Levi, R., Gallo, M. P. & Querio, G. (2020). Squalene: more than a step toward sterols. Antioxidants, 9(8), 688. https://doi.org/10.3390/antiox9080688
Fritsche, S., Wang, X., & Jung, C. (2017). Recent advances in our understanding of tocopherol biosynthesis in plants: an overview of key genes, functions, and breeding of vitamin E improved crops. Antioxidants, 6(4), 99. https://doi.org/10.3390/antiox6040099
Shepherd, T., & Wynne Griffiths, D. (2006). The effects of stresson plant cuticular waxes. New Phytologist, 171(3), 469–499. https://doi.org/10.1111/j.1469-8137.2006.01826.x

Downloads
Published
How to Cite
Issue
Section
Categories
License
Copyright (c) 2026 Creative Science

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.










