Lipid nanoparticles for mR delivery: from rational design and manufacturing to clinical translation
DOI:
https://doi.org/10.55674/cs.v18i2.265802Keywords:
Lipid nanoparticle, mRNA therapeutics, Ionizable cationic lipids, VaccinesAbstract
The clinical success of COVID-19 mRNA vaccines established lipid nanoparticles (LNPs) as a leading platform for in vivo RNA delivery, but their therapeutic potential extends far beyond vaccination. This review provides an integrated perspective on LNP-RNA medicines by linking five interconnected dimensions: historical development, RNA payload engineering, LNP design, manufacturing, and clinical translation. We summarize how nucleoside modification, RNA structural optimization, and emerging formats such as circular RNA and self-amplifying RNA have expanded the functional scope of therapeutic payloads. We then examine the design principles of LNPs, with emphasis on ionizable lipids, formulation composition, and microfluidic manufacturing as the basis for reproducible large-scale production. We further distinguish clinically validated applications, including hepatic RNA delivery and intramuscular mRNA vaccination, from newer but less mature directions such as protein replacement, in vivo gene editing, and in vivo CAR-T generation. Importantly, this review highlights the major barriers that now limit broader translation, including extrahepatic targeting, incomplete understanding of protein corona-mediated delivery, weak cross-species predictability, repeat-dosing challenges, and evolving regulatory requirements. Overall, this review defines the key principles and translational challenges that will shape next-generation LNP-RNA therapeutics.
GRAPHICAL ABSTRACT
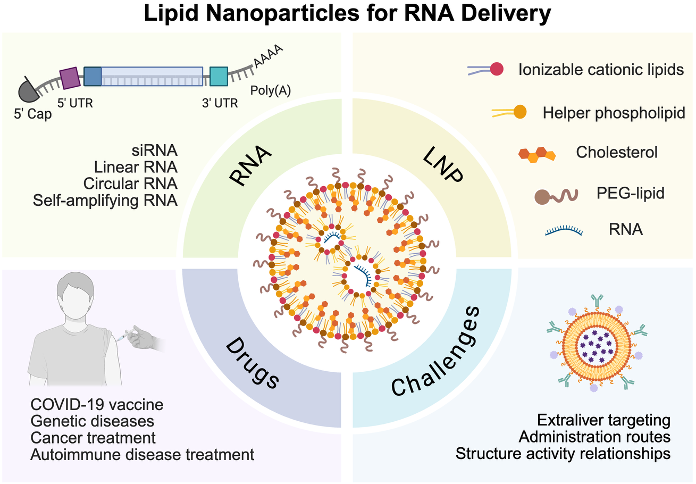
HIGHLIGHTS
- Integrates historical milestones, RNA engineering, LNP design principles, and manufacturing into a unified framework for LNP-RNA therapeutics.
- Introduces clinically validated applications and emerging directions including protein replacement, in vivo gene editing, and in vivo CAR-T therapies.
- Identifies critical translational barriers limiting broader clinical adoption: extrahepatic targeting, incomplete understanding of structure-activity relationships, weak cross-species predictability, and repeat-dosing challenges.
References
Horejs, C. (2021). From lipids to lipid nanoparticles to mRNA vaccines. Nat Rev Mater, 6(12), 1075-1076. https://doi.org/10.1038/s41578-021-00379-9
Brenner, S., Jacob, F., & Meselson, M. (1961). An unstable intermediate carrying information from genes to ribosomes for protein synthesis. Nature, 190, 576-581. https://doi.org/10.1038/190576a0
Gros, F., Hiatt, H., Gilbert, W., Kurland, C. G., Risebrough, R. W., & Watson, J. D. (1961). Unstable ribonucleic acid revealed by pulse labelling of Escherichia coli. Nature, 190, 581-585. https://doi.org/10.1038/190581a0
Wolff, J. A., Malone, R. W., Williams, P., Chong, W., Acsadi, G., Jani, A., & Felgner, P. L. (1990). Direct gene transfer into mouse muscle in vivo. Science, 247(4949 Pt 1), 1465-1468. https://doi.org/10.1126/science.1690918
Fire, A., Xu, S., Montgomery, M. K., Kostas, S. A., Driver, S. E., & Mello, C. C. (1998). Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature, 391(6669), 806-811. https://doi.org/10.1038/35888
Kariko, K., Buckstein, M., Ni, H., & Weissman, D. (2005). Suppression of RNA recognition by Toll-like receptors: the impact of nucleoside modification and the evolutionary origin of RNA. Immunity, 23(2), 165-175. https://doi.org/10.1016/j.immuni.2005.06.008
Kariko, K., Muramatsu, H., Welsh, F. A., Ludwig, J., Kato, H., Akira, S., & Weissman, D. (2008). Incorporation of pseudouridine into mRNA yields superior nonimmunogenic vector with increased translational capacity and biological stability. Mol Ther, 16(11), 1833-1840. https://doi.org/10.1038/mt.2008.200
Bangham, A. D., & Horne, R. W. (1964). Negative Staining of Phospholipids and Their Structural Modification by Surface-Active Agents as Observed in the Electron Microscope. J Mol Biol, 8, 660-668. https://doi.org/10.1016/s0022-2836(64)80115-7
Bangham, A. D., Standish, M. M., & Watkins, J. C. (1965). Diffusion of univalent ions across the lamellae of swollen phospholipids. J Mol Biol, 13(1), 238-252. https://doi.org/10.1016/s0022-2836(65)80093-6
Felgner, P. L., Gadek, T. R., Holm, M., Roman, R., Chan, H. W., Wenz, M., Northrop, J. P., Ringold, G. M., & Danielsen, M. (1987). Lipofection: a highly efficient, lipid-mediated DNA-transfection procedure. Proc Natl Acad Sci U S A, 84(21), 7413-7417. https://doi.org/10.1073/pnas.84.21.7413
Malone, R. W., Felgner, P. L., & Verma, I. M. (1989). Cationic liposome-mediated RNA transfection. Proc Natl Acad Sci U S A, 86(16), 6077-6081. https://doi.org/10.1073/pnas.86.16.6077
Whitehead, K. A., Langer, R., & Anderson, D. G. (2009). Knocking down barriers: advances in siRNA delivery. Nat Rev Drug Discov, 8(2), 129-138. https://doi.org/10.1038/nrd2742
Semple, S. C., Akinc, A., Chen, J., Sandhu, A. P., Mui, B. L., Cho, C. K., Sah, D. W., Stebbing, D., Crosley, E. J., Yaworski, E., Hafez, I. M., Dorkin, J. R., Qin, J., Lam, K., Rajeev, K. G., Wong, K. F., Jeffs, L. B., Nechev, L., Eisenhardt, M. L., .., & Hope, M. J. (2010). Rational design of cationic lipids for siRNA delivery. Nat Biotechnol, 28(2), 172-176. https://doi.org/10.1038/nbt.1602
Belliveau, N. M., Huft, J., Lin, P. J., Chen, S., Leung, A. K., Leaver, T. J., Wild, A. W., Lee, J. B., Taylor, R. J., Tam, Y. K., Hansen, C. L., & Cullis, P. R. (2012). Microfluidic Synthesis of Highly Potent Limit-size Lipid Nanoparticles for In Vivo Delivery of siRNA. Mol Ther Nucleic Acids, 1(8), e37. https://doi.org/10.1038/mtna.2012.28
Leung, A. K., Hafez, I. M., Baoukina, S., Belliveau, N. M., Zhigaltsev, I. V., Afshinmanesh, E., Tieleman, D. P., Hansen, C. L., Hope, M. J., & Cullis, P. R. (2012). Lipid Nanoparticles Containing siRNA Synthesized by Microfluidic Mixing Exhibit an Electron-Dense Nanostructured Core. J Phys Chem C Nanomater Interfaces, 116(34), 18440-18450. https://doi.org/10.1021/jp303267y
U.S. Food and Drug Administration, New Class of Drugs Fulfills Promise of RNA-based Medicine. (2018, August 14). New class of drugs fulfills promise of RNA-based medicine. U.S. Food and Drug Administration. https://www.fda.gov/drugs/spotlight-cder-science/new-class-drugs-fulfills-promise-rna-based-medicine
Baden, L. R., El Sahly, H. M., Essink, B., Kotloff, K., Frey, S., Novak, R., Diemert, D., Spector, S. A., Rouphael, N., Creech, C. B., McGettigan, J., Khetan, S., Segall, N., Solis, J., Brosz, A., Fierro, C., Schwartz, H., Neuzil, K., Corey, L.,..., & Group, C. S. (2021). Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N Engl J Med, 384(5), 403-416. https://doi.org/10.1056/NEJMoa2035389
Polack, F. P., Thomas, S. J., Kitchin, N., Absalon, J., Gurtman, A., Lockhart, S., Perez, J. L., Perez Marc, G., Moreira, E. D., Zerbini, C., Bailey, R., Swanson, K. A., Roychoudhury, S., Koury, K., Li, P., Kalina, W. V., Cooper, D., Frenck, R. W., Jr., Hammitt, L. L., ..., & Group, C. C. T. (2020). Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N Engl J Med, 383(27), 2603-2615. https://doi.org/10.1056/NEJMoa2034577
Thomas, S. J., Moreira, E. D., Jr., Kitchin, N., Absalon, J., Gurtman, A., Lockhart, S., Perez, J. L., Perez Marc, G., Polack, F. P., Zerbini, C., Bailey, R., Swanson, K. A., Xu, X., Roychoudhury, S., Koury, K., Bouguermouh, S., Kalina, W. V., Cooper, D., Frenck, R. W., ..., & Group, C. C. T. (2021). Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine through 6 Months. N Engl J Med, 385(19), 1761-1773. https://doi.org/10.1056/NEJMoa2110345
European Medicines Agency. (2025, March 27). Draft guideline on the quality aspects of mRNA vaccines. European Medicines Agency. https://www.ema.europa.eu/system/files/documents/scientific-guideline/draft-guideline-quality-mrna-vaccines-en.pdf
Andries, O., Mc Cafferty, S., De Smedt, S. C., Weiss, R., Sanders, N. N., & Kitada, T. (2015). N(1)-methylpseudouridine-incorporated mRNA outperforms pseudouridine-incorporated mRNA by providing enhanced protein expression and reduced immunogenicity in mammalian cell lines and mice. J Control Release, 217, 337-344. https://doi.org/10.1016/j.jconrel.2015.08.051
Kariko, K., Muramatsu, H., Ludwig, J., & Weissman, D. (2011). Generating the optimal mRNA for therapy: HPLC purification eliminates immune activation and improves translation of nucleoside-modified, protein-encoding mRNA. Nucleic Acids Res, 39(21), e142. https://doi.org/10.1093/nar/gkr695
Kim, K. Q., Burgute, B. D., Tzeng, S. C., Jing, C., Jungers, C., Zhang, J., Yan, L. L., Vierstra, R. D., Djuranovic, S., Evans, B. S., & Zaher, H. S. (2022). N1-methylpseudouridine found within COVID-19 mRNA vaccines produces faithful protein products. Cell Rep, 40(9), 111300. https://doi.org/10.1016/j.celrep.2022.111300
Nance, K. D., & Meier, J. L. (2021). Modifications in an Emergency: The Role of N1-Methylpseudouridine in COVID-19 Vaccines. ACS Cent Sci, 7(5), 748-756. https://doi.org/10.1021/acscentsci.1c00197
Svitkin, Y. V., Cheng, Y. M., Chakraborty, T., Presnyak, V., John, M., & Sonenberg, N. (2017). N1-methyl-pseudouridine in mRNA enhances translation through eIF2alpha-dependent and independent mechanisms by increasing ribosome density. Nucleic Acids Res, 45(10), 6023-6036. https://doi.org/10.1093/nar/gkx135
Nelson, J., Sorensen, E. W., Mintri, S., Rabideau, A. E., Zheng, W., Besin, G., Khatwani, N., Su, S. V., Miracco, E. J., Issa, W. J., Hoge, S., Stanton, M. G., & Joyal, J. L. (2020). Impact of mRNA chemistry and manufacturing process on innate immune activation. Sci Adv, 6(26), eaaz6893. https://doi.org/10.1126/sciadv.aaz6893
Henderson, J. M., Ujita, A., Hill, E., Yousif-Rosales, S., Smith, C., Ko, N., McReynolds, T., Cabral, C. R., Escamilla-Powers, J. R., & Houston, M. E. (2021). Cap 1 Messenger RNA Synthesis with Co-transcriptional CleanCap((R)) Analog by In Vitro Transcription. Curr Protoc, 1(2), e39. https://doi.org/10.1002/cpz1.39
Shanmugasundaram, M., Senthilvelan, A., & Kore, A. R. (2022). Recent Advances in Modified Cap Analogs: Synthesis, Biochemical Properties, and mRNA Based Vaccines. Chem Rec, 22(8), e202200005. https://doi.org/10.1002/tcr.202200005
Pardi, N., Hogan, M. J., Porter, F. W., & Weissman, D. (2018). mRNA vaccines - a new era in vaccinology. Nat Rev Drug Discov, 17(4), 261-279. https://doi.org/10.1038/nrd.2017.243
Castillo-Hair, S., Fedak, S., Wang, B., Linder, J., Havens, K., Certo, M., & Seelig, G. (2024). Optimizing 5'UTRs for mRNA-delivered gene editing using deep learning. Nat Commun, 15(1), 5284. https://doi.org/10.1038/s41467-024-49508-2
Zhang, H., Liu, H., Xu, Y., Huang, H., Liu, Y., Wang, J., Qin, Y., Wang, H., Ma, L., Xun, Z., Hou, X., Lu, T. K., & Cao, J. (2025). Deep generative models design mRNA sequences with enhanced translational capacity and stability. Science, 390(6773), eadr8470. https://doi.org/10.1126/science.adr8470
Thess, A., Grund, S., Mui, B. L., Hope, M. J., Baumhof, P., Fotin-Mleczek, M., & Schlake, T. (2015). Sequence-engineered mRNA Without Chemical Nucleoside Modifications Enables an Effective Protein Therapy in Large Animals. Mol Ther, 23(9), 1456-1464. https://doi.org/10.1038/mt.2015.103
Presnyak, V., Alhusaini, N., Chen, Y. H., Martin, S., Morris, N., Kline, N., Olson, S., Weinberg, D., Baker, K. E., Graveley, B. R., & Coller, J. (2015). Codon optimality is a major determinant of mRNA stability. Cell, 160(6), 1111-1124. https://doi.org/10.1016/j.cell.2015.02.029
Zhang, H., Zhang, L., Lin, A., Xu, C., Li, Z., Liu, K., Liu, B., Ma, X., Zhao, F., Jiang, H., Chen, C., Shen, H., Li, H., Mathews, D. H., Zhang, Y., & Huang, L. (2023). Algorithm for optimized mRNA design improves stability and immunogenicity. Nature, 621(7978), 396-403. https://doi.org/10.1038/s41586-023-06127-z
Fallahpour, A., Gureghian, V., Filion, G. J., Lindner, A. B., & Pandi, A. (2025). CodonTransformer: a multispecies codon optimizer using context-aware neural networks. Nat Commun, 16(1), 3205. https://doi.org/10.1038/s41467-025-58588-7
Li, S., Moayedpour, S., Li, R., Bailey, M., Riahi, S., Kogler-Anele, L., Miladi, M., Miner, J., Pertuy, F., Zheng, D., Wang, J., Balsubramani, A., Tran, K., Zacharia, M., Wu, M., Gu, X., Clinton, R., Asquith, C., Skaleski, J., …, & Jager, S. (2024). CodonBERT large language model for mRNA vaccines. Genome Res, 34(7), 1027-1035. https://doi.org/10.1101/gr.278870.123
Holtkamp, S., Kreiter, S., Selmi, A., Simon, P., Koslowski, M., Huber, C., Tureci, O., & Sahin, U. (2006). Modification of antigen-encoding RNA increases stability, translational efficacy, and T-cell stimulatory capacity of dendritic cells. Blood, 108(13), 4009-4017. https://doi.org/10.1182/blood-2006-04-015024
Biziaev, N., Shuvalov, A., Salman, A., Egorova, T., Shuvalova, E., & Alkalaeva, E. (2024). The impact of mRNA poly(A) tail length on eukaryotic translation stages. Nucleic Acids Res, 52(13), 7792-7808. https://doi.org/10.1093/nar/gkae510
Grier, A. E., Burleigh, S., Sahni, J., Clough, C. A., Cardot, V., Choe, D. C., Krutein, M. C., Rawlings, D. J., Jensen, M. C., Scharenberg, A. M., & Jacoby, K. (2016). pEVL: A Linear Plasmid for Generating mRNA IVT Templates With Extended Encoded Poly(A) Sequences. Mol Ther Nucleic Acids, 5(4), e306. https://doi.org/10.1038/mtna.2016.21
Geall, A. J., Verma, A., Otten, G. R., Shaw, C. A., Hekele, A., Banerjee, K., Cu, Y., Beard, C. W., Brito, L. A., Krucker, T., O'Hagan, D. T., Singh, M., Mason, P. W., Valiante, N. M., Dormitzer, P. R., Barnett, S. W., Rappuoli, R., Ulmer, J. B., & Mandl, C. W. (2012). Nonviral delivery of self-amplifying RNA vaccines. Proc Natl Acad Sci U S A, 109(36), 14604-14609. https://doi.org/10.1073/pnas.1209367109
Wesselhoeft, R. A., Kowalski, P. S., & Anderson, D. G. (2018). Engineering circular RNA for potent and stable translation in eukaryotic cells. Nat Commun, 9(1), 2629. https://doi.org/10.1038/s41467-018-05096-6
Zhang, J., & Chen, J. (2022). Circular mRNAs: More stable RNAs lead to more persistent protein expression. Mol Ther Nucleic Acids, 30, 357-358. https://doi.org/10.1016/j.omtn.2022.10.014
Zhao, Y. Y., Zhu, F. M., Zhang, Y. J., & Wei, H. Y. (2025). Harnessing the Loop: The Perspective of Circular RNA in Modern Therapeutics. Vaccines (Basel), 13(8), 821. https://doi.org/10.3390/vaccines13080821
Vogel, A. B., Lambert, L., Kinnear, E., Busse, D., Erbar, S., Reuter, K. C., Wicke, L., Perkovic, M., Beissert, T., Haas, H., Reece, S. T., Sahin, U., & Tregoning, J. S. (2018). Self-Amplifying RNA Vaccines Give Equivalent Protection against Influenza to mRNA Vaccines but at Much Lower Doses. Mol Ther, 26(2), 446-455. https://doi.org/10.1016/j.ymthe.2017.11.017
Casmil, I. C., Jin, J., Won, E. J., Huang, C., Liao, S., Cha-Molstad, H., & Blakney, A. K. (2025). The advent of clinical self-amplifying RNA vaccines. Mol Ther, 33(6), 2565-2582. https://doi.org/10.1016/j.ymthe.2025.03.060
Hassett, K. J., Benenato, K. E., Jacquinet, E., Lee, A., Woods, A., Yuzhakov, O., Himansu, S., Deterling, J., Geilich, B. M., Ketova, T., Mihai, C., Lynn, A., McFadyen, I., Moore, M. J., Senn, J. J., Stanton, M. G., Almarsson, O., Ciaramella, G., & Brito, L. A. (2019). Optimization of Lipid Nanoparticles for Intramuscular Administration of mRNA Vaccines. Mol Ther Nucleic Acids, 15, 1-11. https://doi.org/10.1016/j.omtn.2019.01.013
Maier, M. A., Jayaraman, M., Matsuda, S., Liu, J., Barros, S., Querbes, W., Tam, Y. K., Ansell, S. M., Kumar, V., Qin, J., Zhang, X., Wang, Q., Panesar, S., Hutabarat, R., Carioto, M., Hettinger, J., Kandasamy, P., Butler, D., Rajeev, K. G.,..., & Akinc, A. (2013). Biodegradable lipids enabling rapidly eliminated lipid nanoparticles for systemic delivery of RNAi therapeutics. Mol Ther, 21(8), 1570-1578. https://doi.org/10.1038/mt.2013.124
Kulkarni, J. A., Witzigmann, D., Leung, J., Tam, Y. Y. C., & Cullis, P. R. (2019). On the role of helper lipids in lipid nanoparticle formulations of siRNA. Nanoscale, 11(45), 21733-21739. https://doi.org/10.1039/c9nr09347h
Hald Albertsen, C., Kulkarni, J. A., Witzigmann, D., Lind, M., Petersson, K., & Simonsen, J. B. (2022). The role of lipid components in lipid nanoparticles for vaccines and gene therapy. Adv Drug Deliv Rev, 188, 114416. https://doi.org/10.1016/j.addr.2022.114416
Patel, S., Ashwanikumar, N., Robinson, E., DuRoss, A., Sun, C., Murphy-Benenato, K. E., Mihai, C., Almarsson, O., & Sahay, G. (2017). Boosting Intracellular Delivery of Lipid Nanoparticle-Encapsulated mRNA. Nano Lett, 17(9), 5711-5718. https://doi.org/10.1021/acs.nanolett.7b02664
Mui, B. L., Tam, Y. K., Jayaraman, M., Ansell, S. M., Du, X., Tam, Y. Y., Lin, P. J., Chen, S., Narayanannair, J. K., Rajeev, K. G., Manoharan, M., Akinc, A., Maier, M. A., Cullis, P., Madden, T. D., & Hope, M. J. (2013). Influence of Polyethylene Glycol Lipid Desorption Rates on Pharmacokinetics and Pharmacodynamics of siRNA Lipid Nanoparticles. Mol Ther Nucleic Acids, 2(12), e139. https://doi.org/10.1038/mtna.2013.66
Robinson, J. J., Zhang, D., Basak, P., Vaidya, A., Chatterjee, S., Bian, X., Kim, M., Lian, X., Sun, Y., Guerrero, E., Wang, X., Lee, S. M., Liu, S., Gong, J., Tiwari, M., Babanyinah, G. K., Stefan, M. C., Farbiak, L., & Siegwart, D. J. (2025). Reducing Complexity in Lipid Nanoparticles: Three-Component Zwitterionic Amino Lipids for Targeted Extrahepatic mRNA Delivery. ACS Biomater Sci Eng, 11(8), 4853-4868. https://doi.org/10.1021/acsbiomaterials.5c00631
Teng, Y., Guo, Y., Liu, Z., Tang, M., Stewart, W., Xu, X., & Zhang, X. Q. (2025). Computationally Aided Design of Ionizable Cholesteryl Lipids for Lipid Nanoparticles to Modulate Hepatic mRNA Accumulation. J Am Chem Soc, 147(42), 38865-38878. https://doi.org/10.1021/jacs.5c14870
Zhang, H., Liu, D., Yang, K., Liang, Z., & Li, M. (2025). Ionizable guanidine-based lipid nanoparticle for targeted mRNA delivery and cancer immunotherapy. Sci Adv, 11(43), eadx5970. https://doi.org/10.1126/sciadv.adx5970
Fei, Y., Yu, X., Liu, P., Ren, H., Wei, T., & Cheng, Q. (2024). Simplified Lipid Nanoparticles for Tissue- And Cell-Targeted mRNA Delivery Facilitate Precision Tumor Therapy in a Lung Metastasis Mouse Model. Adv Mater, 36(48), e2409812. https://doi.org/10.1002/adma.202409812
Shi, L., Li, Y., Huang, S., Peng, J., Liu, C., Hu, Z., Deng, W., & Deng, G. (2025). mRNA vaccine developed for sequential selective organ-to-cell targeting of glioma. Nat Commun, 17(1), 687. https://doi.org/10.1038/s41467-025-67331-1
Wang, X., Liu, S., Sun, Y., Yu, X., Lee, S. M., Cheng, Q., Wei, T., Gong, J., Robinson, J., Zhang, D., Lian, X., Basak, P., & Siegwart, D. J. (2023). Preparation of selective organ-targeting (SORT) lipid nanoparticles (LNPs) using multiple technical methods for tissue-specific mRNA delivery. Nat Protoc, 18(1), 265-291. https://doi.org/10.1038/s41596-022-00755-x
Roces, C. B., Lou, G., Jain, N., Abraham, S., Thomas, A., Halbert, G. W., & Perrie, Y. (2020). Manufacturing Considerations for the Development of Lipid Nanoparticles Using Microfluidics. Pharmaceutics, 12(11), 1095. https://doi.org/10.3390/pharmaceutics12111095
Zhigaltsev, I. V., Belliveau, N., Hafez, I., Leung, A. K., Huft, J., Hansen, C., & Cullis, P. R. (2012). Bottom-up design and synthesis of limit size lipid nanoparticle systems with aqueous and triglyceride cores using millisecond microfluidic mixing. Langmuir, 28(7), 3633-3640. https://doi.org/10.1021/la204833h
Adams, D., Gonzalez-Duarte, A., O'Riordan, W. D., Yang, C. C., Ueda, M., Kristen, A. V., Tournev, I., Schmidt, H. H., Coelho, T., Berk, J. L., Lin, K. P., Vita, G., Attarian, S., Plante-Bordeneuve, V., Mezei, M. M., Campistol, J. M., Buades, J., Brannagan, T. H., Kim, B. J., …., & Suhr, O. B. (2018). Patisiran, an RNAi Therapeutic, for Hereditary Transthyretin Amyloidosis. N Engl J Med, 379(1), 11-21. https://doi.org/10.1056/NEJMoa1716153
Wilson, E., Goswami, J., Baqui, A. H., Doreski, P. A., Perez-Marc, G., Zaman, K., Monroy, J., Duncan, C. J. A., Ujiie, M., Ramet, M., Perez-Breva, L., Falsey, A. R., Walsh, E. E., Dhar, R., Wilson, L., Du, J., Ghaswalla, P., Kapoor, A., Lan, L.,..., & Conquer, R. S. V. S. G. (2023). Efficacy and Safety of an mRNA-Based RSV PreF Vaccine in Older Adults. N Engl J Med, 389(24), 2233-2244. https://doi.org/10.1056/NEJMoa2307079
Weber, J. S., Carlino, M. S., Khattak, A., Meniawy, T., Ansstas, G., Taylor, M. H., Kim, K. B., McKean, M., Long, G. V., Sullivan, R. J., Faries, M., Tran, T. T., Cowey, C. L., Pecora, A., Shaheen, M., Segar, J., Medina, T., Atkinson, V., Gibney, G. T., …, & Zaks, T. (2024). Individualised neoantigen therapy mRNA-4157 (V940) plus pembrolizumab versus pembrolizumab monotherapy in resected melanoma (KEYNOTE-942): a randomised, phase 2b study. Lancet, 403(10427), 632-644. https://doi.org/10.1016/S0140-6736(23)02268-7
Yap, T. A., Hernando-Calvo, A. H., Calvo, E., Moreno, V., Marquez, R., Papadopoulos, K. P., Garcia-Corbacho, J. G., Hernandez Guerrero, T., Spira, A. I., Tan, D. S. P., Ottensmeier, C. H. H., Koseoglu, S., Chang, P. K., Zhang, E., Martinez, J. L., Trück, C., Magenheim, R., Celik, I., Türeci, Ö., …, & Sahin, U. (2025). First-in-human phase I/II trial evaluating BNT142, a first-in-class mRNA encoded, bispecific antibody targeting Claudin 6 (CLDN6) and CD3, in patients (pts) with CLDN6-positive advanced solid tumors. J Clin Oncol., 43, 2501. https://doi.org/10.1200/JCO.2025.43.16_suppl.2501.
Koeberl, D., Schulze, A., Sondheimer, N., Lipshutz, G. S., Geberhiwot, T., Li, L., Saini, R., Luo, J., Sikirica, V., Jin, L., Liang, M., Leuchars, M., & Grunewald, S. (2024). Interim analyses of a first-in-human phase 1/2 mRNA trial for propionic acidaemia. Nature, 628(8009), 872-877. https://doi.org/10.1038/s41586-024-07266-7
Baek, R., Coughlan, K., Jiang, L., Liang, M., Ci, L., Singh, H., Zhang, H., Kaushal, N., Rajlic, I. L., Van, L., Dimen, R., Cavedon, A., Yin, L., Rice, L., Frassetto, A., Guey, L., Finn, P., & Martini, P. G. V. (2024). Characterizing the mechanism of action for mRNA therapeutics for the treatment of propionic acidemia, methylmalonic acidemia, and phenylketonuria. Nat Commun, 15(1), 3804. https://doi.org/10.1038/s41467-024-47460-9
Gillmore, J. D., Gane, E., Taubel, J., Kao, J., Fontana, M., Maitland, M. L., Seitzer, J., O'Connell, D., Walsh, K. R., Wood, K., Phillips, J., Xu, Y., Amaral, A., Boyd, A. P., Cehelsky, J. E., McKee, M. D., Schiermeier, A., Harari, O., Murphy, A.,..., & Lebwohl, D. (2021). CRISPR-Cas9 In Vivo Gene Editing for Transthyretin Amyloidosis. N Engl J Med, 385(6), 493-502. https://doi.org/10.1056/NEJMoa2107454
Cohn, D. M., Gurugama, P., Magerl, M., Katelaris, C. H., Launay, D., Bouillet, L., Petersen, R. S., Lindsay, K., Aygoren-Pursun, E., Maag, D., Butler, J. S., Shah, M. Y., Golden, A., Xu, Y., Abdelhady, A. M., Lebwohl, D., & Longhurst, H. J. (2025). CRISPR-Based Therapy for Hereditary Angioedema. N Engl J Med, 392(5), 458-467. https://doi.org/10.1056/NEJMoa2405734
Longhurst, H. J., Lindsay, K., Petersen, R. S., Fijen, L. M., Gurugama, P., Maag, D., Butler, J. S., Shah, M. Y., Golden, A., Xu, Y., Boiselle, C., Vogel, J. D., Abdelhady, A. M., Maitland, M. L., McKee, M. D., Seitzer, J., Han, B. W., Soukamneuth, S., Leonard, J., …., & Cohn, D. M. (2024). CRISPR-Cas9 In Vivo Gene Editing of KLKB1 for Hereditary Angioedema. N Engl J Med, 390(5), 432-441. https://doi.org/10.1056/NEJMoa2309149
Musunuru, K., Chadwick, A. C., Mizoguchi, T., Garcia, S. P., DeNizio, J. E., Reiss, C. W., Wang, K., Iyer, S., Dutta, C., Clendaniel, V., Amaonye, M., Beach, A., Berth, K., Biswas, S., Braun, M. C., Chen, H. M., Colace, T. V., Ganey, J. D., Gangopadhyay, S. A.,..., & Kathiresan, S. (2021). In vivo CRISPR base editing of PCSK9 durably lowers cholesterol in primates. Nature, 593(7859), 429-434. https://doi.org/10.1038/s41586-021-03534-y
Rothgangl, T., Dennis, M. K., Lin, P. J. C., Oka, R., Witzigmann, D., Villiger, L., Qi, W., Hruzova, M., Kissling, L., Lenggenhager, D., Borrelli, C., Egli, S., Frey, N., Bakker, N., Walker, J. A., Kadina, A. P., Victorov, D. V., Pacesa, M., Kreutzer, S., …., & Schwank, G. (2021). In vivo adenine base editing of PCSK9 in macaques reduces LDL cholesterol levels. Nat Biotechnol, 39(8), 949-957. https://doi.org/10.1038/s41587-021-00933-4
Tremblay, F., Xiong, Q., Shah, S. S., Ko, C. W., Kelly, K., Morrison, M. S., Giancarlo, C., Ramirez, R. N., Hildebrand, E. M., Voytek, S. B., El Sebae, G. K., Wright, S. H., Lofgren, L., Clarkson, S., Waters, C., Linder, S. J., Liu, S., Eom, T., Parikh, S., …., & Jaffe, A. B. (2025). A potent epigenetic editor targeting human PCSK9 for durable reduction of low-density lipoprotein cholesterol levels. Nat Med, 31(4), 1329-1338. https://doi.org/10.1038/s41591-025-03508-x
Mougiakakos, D., Kronke, G., Volkl, S., Kretschmann, S., Aigner, M., Kharboutli, S., Boltz, S., Manger, B., Mackensen, A., & Schett, G. (2021). CD19-Targeted CAR T Cells in Refractory Systemic Lupus Erythematosus. N Engl J Med, 385(6), 567-569. https://doi.org/10.1056/NEJMc2107725
Rurik, J. G., Tombacz, I., Yadegari, A., Mendez Fernandez, P. O., Shewale, S. V., Li, L., Kimura, T., Soliman, O. Y., Papp, T. E., Tam, Y. K., Mui, B. L., Albelda, S. M., Pure, E., June, C. H., Aghajanian, H., Weissman, D., Parhiz, H., & Epstein, J. A. (2022). CAR T cells produced in vivo to treat cardiac injury. Science, 375(6576), 91-96. https://doi.org/10.1126/science.abm0594
CapstanTherapeutics. (2025). A study of CPTX2309 in healthy participants (ClinicalTrials.gov Identifier: NCT06917742)[Clinical trial registration].
Wang, Q., Xiao, Z. X., Zheng, X., Wang, G., Yang, L., Shi, L., Xiang, N., Wang, X., Zha, G. F., Schett, G., & Chen, Z. (2025). In Vivo CD19 CAR T-Cell Therapy for Refractory Systemic Lupus Erythematosus. N Engl J Med, 393(15), 1542-1544. https://doi.org/10.1056/NEJMc2509522
O'Leary, E., Jiang, Y., Kristensen, L. S., Hansen, T. B., & Kjems, J. (2025). The therapeutic potential of circular RNAs. Nat Rev Genet, 26(4), 230-244. https://doi.org/10.1038/s41576-024-00806-x
Wayne, C. J., & Blakney, A. K. (2024). Self-amplifying RNA COVID-19 vaccine. Cell, 187(8), 1822-1822 e1821. https://doi.org/10.1016/j.cell.2024.03.018
Omata, D., Kawahara, E., Munakata, L., Tanaka, H., Akita, H., Yoshioka, Y., & Suzuki, R. (2024). Effect of Anti-PEG Antibody on Immune Response of mRNA-Loaded Lipid Nanoparticles. Mol Pharm, 21(11), 5672-5680. https://doi.org/10.1021/acs.molpharmaceut.4c00628
Luozhong, S., Liu, P., Li, R., Yuan, Z., Debley, E., Chen, Y., Hu, Y., Cao, Z., Cui, M., McIlhenny, K., McCurdy, C., Bhashyam, D., Wilkens, S., Zhang, P., Kwan, A., Grossman, M., Lai, R., Ma, Y., Lipkin, S., & Jiang, S. (2025). Poly(carboxybetaine) lipids enhance mRNA therapeutics efficacy and reduce their immunogenicity. Nat Mater, 24(11), 1852-1861. https://doi.org/10.1038/s41563-025-02240-8
Cheng, Q., Wei, T., Farbiak, L., Johnson, L. T., Dilliard, S. A., & Siegwart, D. J. (2020). Selective organ targeting (SORT) nanoparticles for tissue-specific mRNA delivery and CRISPR-Cas gene editing. Nat Nanotechnol, 15(4), 313-320. https://doi.org/10.1038/s41565-020-0669-6
Dilliard, S. A., Cheng, Q., & Siegwart, D. J. (2021). On the mechanism of tissue-specific mRNA delivery by selective organ targeting nanoparticles. Proc Natl Acad Sci U S A, 118(52), e2109256118. https://doi.org/10.1073/pnas.2109256118
Dilliard, S. A., & Siegwart, D. J. (2023). Passive, active and endogenous organ-targeted lipid and polymer nanoparticles for delivery of genetic drugs. Nat Rev Mater, 8(4), 282-300. https://doi.org/10.1038/s41578-022-00529-7
Odunze, U., Rustogi, N., Devine, P., Miller, L., Pereira, S., Vashist, S., Snijder, H. J., Corkill, D., Sabirsh, A., Douthwaite, J., Bond, N., & Desai, A. (2024). RNA encoded peptide barcodes enable efficient in vivo screening of RNA delivery systems. Nucleic Acids Res, 52(16), 9384-9396. https://doi.org/10.1093/nar/gkae648
Sago, C. D., Lokugamage, M. P., Paunovska, K., Vanover, D. A., Monaco, C. M., Shah, N. N., Gamboa Castro, M., Anderson, S. E., Rudoltz, T. G., Lando, G. N., Munnilal Tiwari, P., Kirschman, J. L., Willett, N., Jang, Y. C., Santangelo, P. J., Bryksin, A. V., & Dahlman, J. E. (2018). High-throughput in vivo screen of functional mRNA delivery identifies nanoparticles for endothelial cell gene editing. Proc Natl Acad Sci U S A, 115(42), E9944-E9952. https://doi.org/10.1073/pnas.1811276115
Wang, L., Zhou, W., Chen, H., Jia, X., Zheng, P., Jiang, H., Wu, M., Zhang, Y., Ding, Y., Peng, Y., Zhu, R., Li, T., Tian, B., Du, B., & Du, J. (2025). Barcoded screening identifies nanocarriers for protein delivery to kidney. Nat Commun, 16(1), 899. https://doi.org/10.1038/s41467-025-56257-3
Hofstraat, S. R. J., Anbergen, T., Zwolsman, R., Deckers, J., van Elsas, Y., Trines, M. M., Versteeg, I., Hoorn, D., Ros, G. W. B., Bartelet, B. M., Hendrikx, M. M. A., Darwish, Y. B., Kleuskens, T., Borges, F., Maas, R. J. F., Verhalle, L. M., Tielemans, W., Vader, P., de Jong, O. G.,..., & van der Meel, R. (2025). Nature-inspired platform nanotechnology for RNA delivery to myeloid cells and their bone marrow progenitors. Nat Nanotechnol, 20(4), 532-542. https://doi.org/10.1038/s41565-024-01847-3
Liu, S., Zhang, L., Wu, W., Chen, M. Y., & Qian, F. (2026). Spleen-targeted mRNA delivery via long-chain PEGylated lipids at low molar ratio enhances antitumor immunity against melanoma. Mol Ther Nucleic Acids, 37(1), 102803. https://doi.org/10.1016/j.omtn.2025.102803
Qiu, M., Tang, Y., Chen, J., Muriph, R., Ye, Z., Huang, C., Evans, J., Henske, E. P., & Xu, Q. (2022). Lung-selective mRNA delivery of synthetic lipid nanoparticles for the treatment of pulmonary lymphangioleiomyomatosis. Proc Natl Acad Sci U S A, 119(8), e2116271119. https://doi.org/10.1073/pnas.2116271119
Somu Naidu, G., Rampado, R., Sharma, P., Ezra, A., Kundoor, G. R., Breier, D., & Peer, D. (2025). Ionizable Lipids with Optimized Linkers Enable Lung-Specific, Lipid Nanoparticle-Mediated mRNA Delivery for Treatment of Metastatic Lung Tumors. ACS Nano, 19(6), 6571-6587. https://doi.org/10.1021/acsnano.4c18636
Zhang, Y., Shao, B., Jiang, K., Cen, J., Liu, Y., Tan, J., Zhang, L., Li, K., You, C., He, Z., Zhang, G., Chen, M., Hu, J., & Liu, S. (2025). Fluorinated Ionizable Lipids for Efficient Spleen-Targeted mRNA Delivery in Cancer Immunotherapy. J Am Chem Soc, 147(45), 41756-41767. https://doi.org/10.1021/jacs.5c13982
Breda, L., Papp, T. E., Triebwasser, M. P., Yadegari, A., Fedorky, M. T., Tanaka, N., Abdulmalik, O., Pavani, G., Wang, Y., Grupp, S. A., Chou, S. T., Ni, H., Mui, B. L., Tam, Y. K., Weissman, D., Rivella, S., & Parhiz, H. (2023). In vivo hematopoietic stem cell modification by mRNA delivery. Science, 381(6656), 436-443. https://doi.org/10.1126/science.ade6967
Tarab-Ravski, D., Hazan-Halevy, I., Goldsmith, M., Stotsky-Oterin, L., Breier, D., Naidu, G. S., Aitha, A., Diesendruck, Y., Ng, B. D., Barsheshet, H., Berger, T., Vaxman, I., Raanani, P., & Peer, D. (2023). Delivery of Therapeutic RNA to the Bone Marrow in Multiple Myeloma Using CD38-Targeted Lipid Nanoparticles. Adv Sci (Weinh), 10(21), e2301377. https://doi.org/10.1002/advs.202301377
Tombacz, I., Laczko, D., Shahnawaz, H., Muramatsu, H., Natesan, A., Yadegari, A., Papp, T. E., Alameh, M. G., Shuvaev, V., Mui, B. L., Tam, Y. K., Muzykantov, V., Pardi, N., Weissman, D., & Parhiz, H. (2021). Highly efficient CD4+ T cell targeting and genetic recombination using engineered CD4+ cell-homing mRNA-LNPs. Mol Ther, 29(11), 3293-3304. https://doi.org/10.1016/j.ymthe.2021.06.004
Herrera-Barrera, M., Ryals, R. C., Gautam, M., Jozic, A., Landry, M., Korzun, T., Gupta, M., Acosta, C., Stoddard, J., Reynaga, R., Tschetter, W., Jacomino, N., Taratula, O., Sun, C., Lauer, A. K., Neuringer, M., & Sahay, G. (2023). Peptide-guided lipid nanoparticles deliver mRNA to the neural retina of rodents and nonhuman primates. Sci Adv, 9(2), eadd4623. https://doi.org/10.1126/sciadv.add4623
Luozhong, S., Yuan, Z., Sarmiento, T., Chen, Y., Gu, W., McCurdy, C., Gao, W., Li, R., Wilkens, S., & Jiang, S. (2022). Phosphatidylserine Lipid Nanoparticles Promote Systemic RNA Delivery to Secondary Lymphoid Organs. Nano Lett, 22(20), 8304-8311. https://doi.org/10.1021/acs.nanolett.2c03234
Papp, T. E., Zeng, J., Shahnawaz, H., Akyianu, A., Breda, L., Yadegari, A., Steward, J., Shi, R., Li, Q., Mui, B. L., Tam, Y. K., Weissman, D., Rivella, S., Shuvaev, V., Muzykantov, V. R., & Parhiz, H. (2025). CD47 peptide-cloaked lipid nanoparticles promote cell-specific mRNA delivery. Mol Ther, 33(7), 3195-3208. https://doi.org/10.1016/j.ymthe.2025.03.018
Sebastiani, F., Yanez Arteta, M., Lerche, M., Porcar, L., Lang, C., Bragg, R. A., Elmore, C. S., Krishnamurthy, V. R., Russell, R. A., Darwish, T., Pichler, H., Waldie, S., Moulin, M., Haertlein, M., Forsyth, V. T., Lindfors, L., & Cardenas, M. (2021). Apolipoprotein E Binding Drives Structural and Compositional Rearrangement of mRNA-Containing Lipid Nanoparticles. ACS Nano, 15(4), 6709-6722. https://doi.org/10.1021/acsnano.0c10064
Akinc, A., Maier, M. A., Manoharan, M., Fitzgerald, K., Jayaraman, M., Barros, S., Ansell, S., Du, X., Hope, M. J., Madden, T. D., Mui, B. L., Semple, S. C., Tam, Y. K., Ciufolini, M., Witzigmann, D., Kulkarni, J. A., van der Meel, R., & Cullis, P. R. (2019). The Onpattro story and the clinical translation of nanomedicines containing nucleic acid-based drugs. Nat Nanotechnol, 14(12), 1084-1087. https://doi.org/10.1038/s41565-019-0591-y
Liu, X., Campos-Gomez, J., Luo, M., Ma, J., Chen, H. C., Shukla, S., Wu, S., Snow, G., Pei, Y., Boehme, S. A., Geller, D. E., Rowe, S. M., & Engelhardt, J. F. (2025). LUNAR LNP delivery of CFTR mRNA restores channel function and improves mucociliary clearance in ferret cystic fibrosis airways. Mol Ther. https://doi.org/10.1016/j.ymthe.2025.12.040
Robinson, E., MacDonald, K. D., Slaughter, K., McKinney, M., Patel, S., Sun, C., & Sahay, G. (2018). Lipid Nanoparticle-Delivered Chemically Modified mRNA Restores Chloride Secretion in Cystic Fibrosis. Mol Ther, 26(8), 2034-2046. https://doi.org/10.1016/j.ymthe.2018.05.014
Wei, T., Sun, Y., Cheng, Q., Chatterjee, S., Traylor, Z., Johnson, L. T., Coquelin, M. L., Wang, J., Torres, M. J., Lian, X., Wang, X., Xiao, Y., Hodges, C. A., & Siegwart, D. J. (2023). Lung SORT LNPs enable precise homology-directed repair mediated CRISPR/Cas genome correction in cystic fibrosis models. Nat Commun, 14(1), 7322. https://doi.org/10.1038/s41467-023-42948-2
Bai, X., Chen, Q., Li, F., Teng, Y., Tang, M., Huang, J., Xu, X., & Zhang, X. Q. (2024). Optimized inhaled LNP formulation for enhanced treatment of idiopathic pulmonary fibrosis via mRNA-mediated antibody therapy. Nat Commun, 15(1), 6844. https://doi.org/10.1038/s41467-024-51056-8
Liu, S., Wen, Y., Shan, X., Ma, X., Yang, C., Cheng, X., Zhao, Y., Li, J., Mi, S., Huo, H., Li, W., Jiang, Z., Li, Y., Lin, J., Miao, L., & Lu, X. (2024). Charge-assisted stabilization of lipid nanoparticles enables inhaled mRNA delivery for mucosal vaccination. Nat Commun, 15(1), 9471. https://doi.org/10.1038/s41467-024-53914-x
Ball, R. L., Bajaj, P., & Whitehead, K. A. (2018). Oral delivery of siRNA lipid nanoparticles: Fate in the GI tract. Sci Rep, 8(1), 2178. https://doi.org/10.1038/s41598-018-20632-6
Sung, J., Alghoul, Z., Long, D., Yang, C., & Merlin, D. (2022). Oral delivery of IL-22 mRNA-loaded lipid nanoparticles targeting the injured intestinal mucosa: A novel therapeutic solution to treat ulcerative colitis. Biomaterials, 288, 121707. https://doi.org/10.1016/j.biomaterials.2022.121707
Suri, K., Pfeifer, L., Cvet, D., Li, A., McCoy, M., Singh, A., & Amiji, M. M. (2025). Oral delivery of stabilized lipid nanoparticles for nucleic acid therapeutics. Drug Deliv Transl Res, 15(5), 1755-1769. https://doi.org/10.1007/s13346-024-01709-4
Huang, X., Liu, C., Sharma, S. N., You, X., Chen, S., Li, Y., Liu, H. J., Liu, B., Saiding, Q., Chen, W., Lee, Y., Kong, N., Abdi, R., & Tao, W. (2025). Oral delivery of liquid mRNA therapeutics by an engineered capsule for treatment of preclinical intestinal disease. Sci Transl Med, 17(807), eadu1493. https://doi.org/10.1126/scitranslmed.adu1493
Ma, T., Wang, L., Yang, T., Ma, G., & Wang, S. (2014). M-cell targeted polymeric lipid nanoparticles containing a Toll-like receptor agonist to boost oral immunity. Int J Pharm, 473(1-2), 296-303. https://doi.org/10.1016/j.ijpharm.2014.06.052
Di Trani, C. A., Cirella, A., Arrizabalaga, L., Alvarez, M., Bella, A., Fernandez-Sendin, M., Russo-Cabrera, J. S., Gomar, C., Ardaiz, N., Teijeira, A., Bolanos, E., Gonzalez-Gomariz, J., Otano, I., Aranda, F., Palencia, B., Segues, A., Huang, S., van Duijnhoven, S. M. J., van Elsas, A.,…, & Berraondo, P. (2023). Intratumoral injection of IL-12-encoding mRNA targeted to CSFR1 and PD-L1 exerts potent anti-tumor effects without substantial systemic exposure. Mol Ther Nucleic Acids, 33, 599-616. https://doi.org/10.1016/j.omtn.2023.07.020
Hewitt, S. L., Bailey, D., Zielinski, J., Apte, A., Musenge, F., Karp, R., Burke, S., Garcon, F., Mishra, A., Gurumurthy, S., Watkins, A., Arnold, K., Moynihan, J., Clancy-Thompson, E., Mulgrew, K., Adjei, G., Deschler, K., Potz, D., Moody, G.,..., & Luheshi, N. (2020). Intratumoral IL12 mRNA Therapy Promotes TH1 Transformation of the Tumor Microenvironment. Clin Cancer Res, 26(23), 6284-6298. https://doi.org/10.1158/1078-0432.CCR-20-0472
Sun, M., Ma, B., Pan, Z., Zhao, Y., Tian, L., Fan, Y., Kong, W., Wang, J., Xu, B., Ao, Y., Guo, Q., Wang, X., Peng, X., Li, X., Cheng, J., Miao, L., Wang, K., & Hu, X. (2024). Targeted Therapy of Osteoarthritis via Intra-Articular Delivery of Lipid-Nanoparticle-Encapsulated Recombinant Human FGF18 mRNA. Adv Healthc Mater, 13(29), e2400804. https://doi.org/10.1002/adhm.202400804
Paunovska, K., Sago, C. D., Monaco, C. M., Hudson, W. H., Castro, M. G., Rudoltz, T. G., Kalathoor, S., Vanover, D. A., Santangelo, P. J., Ahmed, R., Bryksin, A. V., & Dahlman, J. E. (2018). A Direct Comparison of in Vitro and in Vivo Nucleic Acid Delivery Mediated by Hundreds of Nanoparticles Reveals a Weak Correlation. Nano Lett, 18(3), 2148-2157. https://doi.org/10.1021/acs.nanolett.8b00432
Paunovska, K., Loughrey, D., & Dahlman, J. E. (2022). Drug delivery systems for RNA therapeutics. Nat Rev Genet, 23(5), 265-280. https://doi.org/10.1038/s41576-021-00439-4

Downloads
Published
How to Cite
Issue
Section
Categories
License
Copyright (c) 2026 Creative Science

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.










