Effects of soil properties and heavy metals with a bioactive compound, acteoside, in Acanthus ebracteatus from three different habitats of eastern part of Thailand
DOI:
https://doi.org/10.55674/cs.v18i2.265557Keywords:
Acanthus ebracteatus, acteoside, soil properties, heavy metal accumulationAbstract
Acanthus ebracteatus Vahl is a mangrove medicinal plant widely distributed along the coastal regions of Thailand. It is recognized as a rich natural source of acteoside, a phenylethanoid glycoside and is discovered in the anti-inflammatory, antioxidant, and hepatoprotective activities. However, the information availability regarding the influence of environmental factors, particularly soil properties and heavy metal accumulation, on acteoside biosynthesis is limited. The aims of this study were to investigate the effects of soil physicochemical characteristics and heavy metal concentrations on acteoside content in A. ebracteatus collected from three distinct habitats in eastern Thailand: Chachoengsao, Chonburi, and Rayong. Soil samples were analyzed for pH, organic matter, and macronutrient contents, including nitrogen (N), and phosphorus (P), as well as concentrations of As, Cd, Hg, and Pb using inductively coupled plasma – optical emission spectrometer (ICP–OES). Acteoside levels in methanolic leaf extracts were quantified by high-performance liquid chromatography coupled with diode-array detection (HPLC-DAD). Correlations between soil parameters and acteoside content were evaluated. Based on our searching literature found that an association between soil heavy metal levels and acteoside concentration in A. ebracteatus is demonstrated. The results revealed significant variations in soil composition among the sampling sites. The highest acteoside accumulation was observed in plants from the Chachoengsao habitat, which was characterized by near-neutral soil pH (7.1 ± 0.2) and relatively elevated concentrations of Hg (0.06 ± 0.00 mg kg⁻¹) and Pb (20.07 ± 0.81 mg kg⁻¹). A strong positive correlation was identified between acteoside content and soil Hg and Pb concentrations. These findings indicated that soil mineral composition and heavy metal availability played a crucial role in modulating acteoside biosynthesis in A. ebracteatus. The results provide valuable insights for phytochemical quality control, environmental monitoring, and the sustainable cultivation of this medicinal plant within the Eastern Economic Corridor (EEC) of Thailand.
GRAPHICAL ABSTRACT
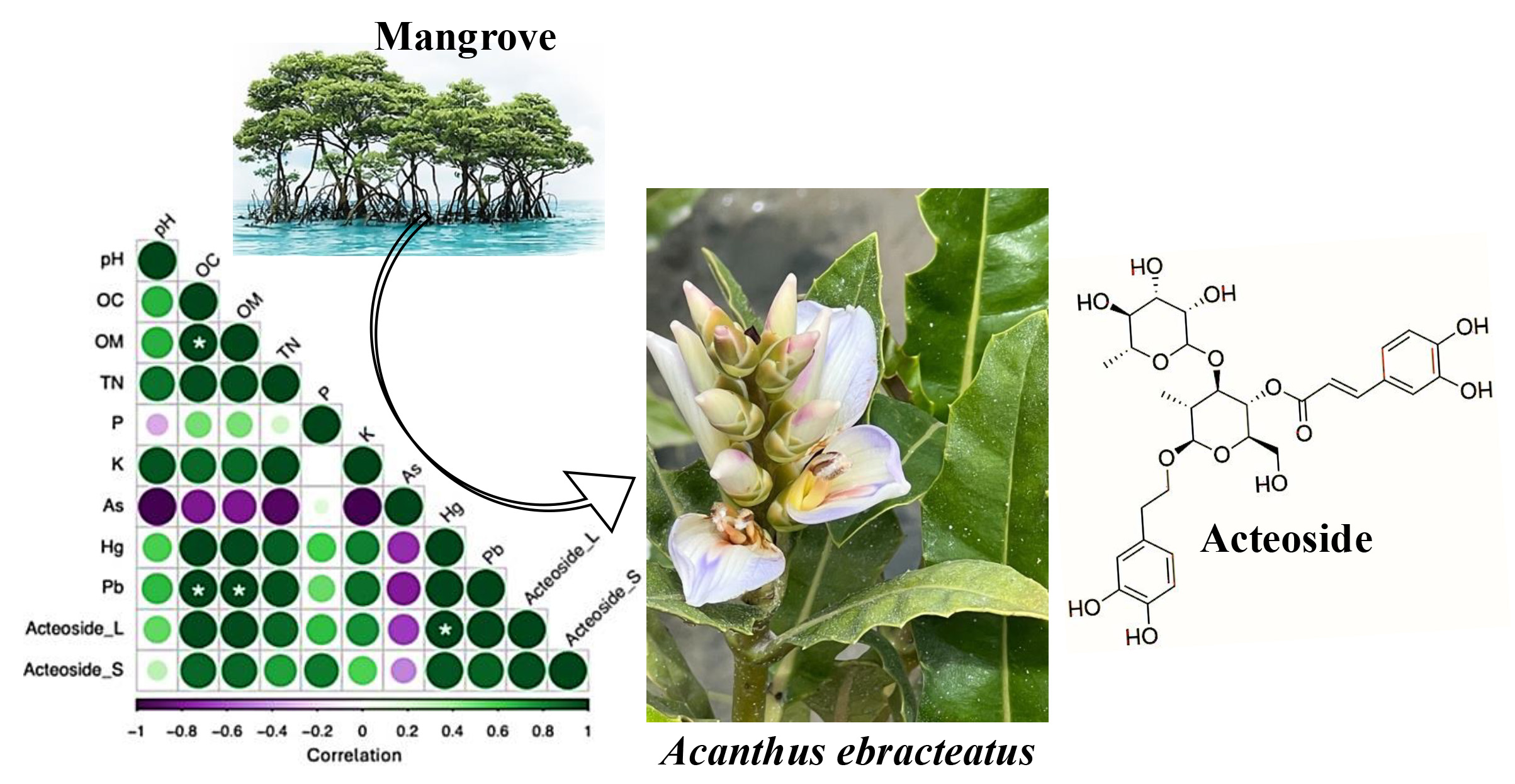
HIGHLIGHTS
- Soil physicochemical properties and heavy metal levels significantly influence acteoside accumulation in Acanthus ebracteatus across different eastern Thai habitats.
- Plants from Chachoengsao exhibited the highest acteoside content, strongly correlated with near-neutral soil pH and elevated Hg and Pb concentrations.
- These findings provide important insights for phytochemical quality control and support sustainable cultivation strategies for medicinal mangrove plants in Thailand’s Eastern Economic Corridor.
References
Li, Q., Cai, S., Mo, C., Chu, B., Peng, L., & Yang, F. (2010). Toxic effects of heavy metals and their accumulation in vegetables grown in a saline soil. Ecotoxicology and Environmental Safety, 73(1), 84-88. https://doi.org/10.1016/j.ecoenv.2009.09.002
Vinogradova, N., Glukhov, A., Chaplygin, V., Kumar, P., Mandzhieva, S., Minkina, T., & Rajput, V. D. (2023). The content of heavy metals in medicinal plants in various environmental conditions: A review. Horticulturae, 9(2), 239. https://doi.org/10.3390/horticulturae9020239
Barthwal, J., Smitha, N. A. I. R., & Kakkar, P. (2008). Heavy metal accumulation in medicinal plants collected from environmentally different sites. Biomedical and environmental sciences, 21(4), 319-324. https://doi.org/10.1016/S0895-3988(08)60049-5
Asiminicesei, D. M., Fertu, D. I., & Gavrilescu, M. (2024). Impact of heavy metal pollution in the environment on the metabolic profile of medicinal plants and their therapeutic potential. Plants, 13(6), 913.https://doi.org/10.3390/plants13060913
Biswas, T., Parveen, O., Pandey, V. P., Mathur, A., & Dwivedi, U. N. (2020). Heavy metal accumulation efficiency, growth and centelloside production in the medicinal herb Centella asiatica (L.) urban under different soil concentrations of cadmium and lead. Industrial Crops and Products, 157, 112948.https://doi.org/10.1016/j.indcrop.2020.112948
Reshi, Z. A., Ahmad, W., Lukatkin, A. S., & Javed, S. B. (2023). From nature to lab: A review of secondary metabolite biosynthetic pathways, environmental influences, and in vitro approaches. Metabolites, 13(8), 895. https://doi.org/10.3390/metabo13080895
Hokputsa, S., Harding, S. E., Inngjerdingen, K., Jumel, K., Michaelsen, T. E., Heinze, T., & Paulsen, B. S. (2004). Bioactive polysaccharides from the stems of the Thai medicinal plant Acanthus ebracteatus: their chemical and physical features. Carbohydrate research, 339(4), 753-762.https://doi.org/10.1016/j.carres.2003.11.022
Olatunji, O. J., Olatunde, O. O., Jayeoye, T. J., Singh, S., Nalinbenjapun, S., Sripetthong, S., & Ovatlarnporn, C. (2022). New insights on Acanthus ebracteatus Vahl: UPLC-ESI-QTOF-MS profile, antioxidant, antimicrobial and anticancer activities. Molecules, 27(6), 1981. https://doi.org/10.3390/molecules27061981
Kanchanapoom, T., Kasai, R., Picheansoonthon, C., & Yamasaki, K. (2001). Megastigmane, aliphatic alcohol and benzoxazinoid glycosides from Acanthus ebracteatus. Phytochemistry, 58(5), 811-817.https://doi.org/10.1016/S0031-9422(01)00306-5
Anh, B. T. M., Nga, T. T. T., Lan, H. T. T., Mai, N. T., Huong, P. T. T., Tai, B. H., & Van Kiem, P. (2023). Phytochemical Constituents from the Aerial Parts of Acanthus ebracteatus Vahl. and Their Cytotoxic Activity. Natural Product Communications, 18(3), 1934578X231166547. https://doi.org/10.1177/1934578X231166547
He, J., Hu, X. P., Zeng, Y., Li, Y., Wu, H. Q., Qiu, R. Z., & He, Z. D. (2011). Advanced research on acteoside for chemistry and bioactivities. Journal of Asian Natural Products Research, 13(5), 449-464.https://doi.org/10.1080/10286020.2011.568940
Kanlayavattanakul, M., Khongkow, M., & Lourith, N. (2024). Wound healing and photoprotection properties of Acanthus ebracteatus Vahl. extracts standardized in verbascoside. Scientific Reports, 14(1), 1904.https://doi.org/10.1038/s41598-024-52511-8
Bureau of Drug and Narcotic. (2022). Thai Herbal Pharmacopoeia 2021 Supplement 2022: Acanthus ebracteatus. Department of Medical Sciences, Ministry of Public Health, Thailand.
Thongra-ar, W., & Parkpianl, P. (2002). Total mercury concentrations in coastal areas of Thailand: A review. ScienceAsia, 28, 301–312.
https:// doi:10.2306/scienceasia1513-1874.2002.28.301
Department of Pollution Control. (2023). Annual Report on Environmental Quality in the Eastern Economic Corridor (EEC). Ministry of Natural Resources and Environment, Thailand.
Thongra-ar, W., Musika, C., Wongsudawan, W., & Munhapol, A. (2008). Heavy metals contamination in sediments along the eastern coast of the gulf of Thailand. Environment Asia, 1(1), 37-45.
Sharma, A., Shahzad, B., Rehman, A., Bhardwaj, R., Landi, M., & Zheng, B. (2019). Response of phenylpropanoid pathway and the role of polyphenols in plants under abiotic stress. Molecules, 24(13), 2452.https://doi.org/10.3390/molecules24132452
Pandey, A., Agrawal, M., & Agrawal, S. B. (2023). Ultraviolet-B and heavy metal-induced regulation of secondary metabolites in medicinal plants: a review. Metabolites, 13(3), 341.https://doi.org/10.3390/metabo13030341
Muscolo, A., Sidari, M., Settineri, G., Papalia, T., Mallamaci, C., & Attinà, E. (2019). Influence of Soil Properties on Bioactive Compounds and Antioxidant Capacity of Brassica rupestris Raf. Journal of Soil Science and Plant Nutrition, 19(4), 808-815.https://doi.org/10.1007/s42729-019-00080-5
Siregar, E. S., Jumilawaty, E., Tanjung, M., Syafitri, A., Kusmana, C., Basyuni, M., Hartanto, A., & Rahmania, R. (2025). Bioaccumulation of heavy metals by Acanthus ilicifolius in polluted mangrove ecosystems. Emerging Science Journal, 9(2), 557–568. https://doi.org/10.28991/ESJ-2025-09-02-03
Ministry of Higher Education, Science, Research and Innovation. (2021). Thailand BCG economy development plan 2021–2027. National Science, Technology and Innovation Policy Office.
Walkley, A., & Black, I. A. (1934). An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Science, 37(1), 29–38.
U.S. Environmental Protection Agency. (2018). Method 6010D: Inductively coupled plasma–optical emission spectrometry (ICP–OES). U.S. EPA.
Wei, T., & Simko, V. (2021). R package “corrplot”: Visualization of a correlation matrix (Version 0.92) [Software]. https://github.com/taiyun/corrplot
Chaouqi, S., Moratalla-López, N., Alonso, G. L., Lorenzo, C., Zouahri, A., Asserar, N., & Guedira, T. (2023). Effect of soil composition on secondary metabolites of Moroccan saffron (Crocus sativus L.). Plants, 12(4), 711.https://doi.org/10.3390/plants12040711
Li, Z., Jiang, H., Yan, H., Jiang, X., Ma, Y., & Qin, Y. (2021). Carbon and nitrogen metabolism under nitrogen variation affects flavonoid accumulation in the leaves of Coreopsis tinctoria. PeerJ, 9, e12152. https://doi.org/10.7717/peerj.12152
Liu, S., Shi, X., Yang, G., Khokiattiwong, S., & Kornkanitnan, N. (2016). Concentration distribution and assessment of heavy metals in the surface sediments of the western Gulf of Thailand. Environmental Earth Sciences, 75(4), 346.https://doi.org/10.1007/s12665-016-5422-y
Jańczak-Pieniążek, M., Cichoński, J., Michalik, P., & Chrzanowski, G. (2022). Effect of heavy metal stress on phenolic compounds accumulation in winter wheat plants. Molecules, 28(1), 241.https://doi.org/10.3390/molecules28010241
MacFarlane, G. R., Pulkownik, A., & Burchett, M. D. (2003). Accumulation and distribution of heavy metals in the grey mangrove, Avicennia marina (Forsk.) Vierh.: biological indication potential. Environmental Pollution, 123(1), 139-151.https://doi.org/10.1016/S0269-7491(02)00342-1
González-Ocampo, H. A., Martínez-Álvarez, I. G., Jaramillo-Flores, M. E., & Luna-González, A. (2022). Comparison of phenolic and flavonoid content and antioxidant and chelating activities of Rhizophora mangle in different anthropogenically-polluted coastal lagoons. Frontiers in Marine Science, 9, 791748.https://doi.org/10.3389/fmars.2022.791748
Salam, U., Ullah, S., Tang, Z. H., Elateeq, A. A., Khan, Y., Khan, J., & Ali, S. (2023). Plant metabolomics: an overview of the role of primary and secondary metabolites against different environmental stress factors. Life, 13(3), 706.https://doi.org/10.3390/life13030706
Alvarez-Rivera, G., Sanz, A., Cifuentes, A., Ibánez, E., Paape, T., Lucas, M. M., & Pueyo, J. J. (2022). Flavonoid accumulation varies in Medicago truncatula in response to mercury stress. Frontiers in Plant Science, 13, 933209.https://doi.org/10.3389/fpls.2022.933209
Piwowarczyk, R., Ochmian, I., Lachowicz, S., Kapusta, I., Malinowska, K., & Ruraż, K. (2021). Correlational nutritional relationships and interactions between expansive holoparasite Orobanche laxissima and woody hosts on metal-rich soils. Phytochemistry, 190, 112844. https://doi.org/10.1016/j.phytochem.2021.112844
Danaeipour, R., & Sharifi, M. (2022). Determination and evaluation of acteoside content of Scrophularia striata Boiss. under lead stress. Medical Sciences Forum, 14(1), 123. https://doi.org/10.3390/ECMC2022-13305
Mohammad, S. I., Kareem, A. K., AlMohamadi, H., Vasudevan, A., Rekha, M. M., Gayathri, S., & Mustafa, Y. F. (2025). Morphophysiological and phytochemical responses to arsenic, cadmium and lead stress in parsley (Petroselinum crispum) [Preprint]. https://doi.org/10.21203/rs.3.rs-7455730/v1

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Creative Science

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.










