Exploring phytochemicals and antidiabetic property of the hexane extract from Abutilon indicum (L.) Sweet roots
DOI:
https://doi.org/10.55674/cs.v18i2.265485Keywords:
Sterol, Sterol glycoside, -glucosidase inhibitory activity, Molecular dockingAbstract
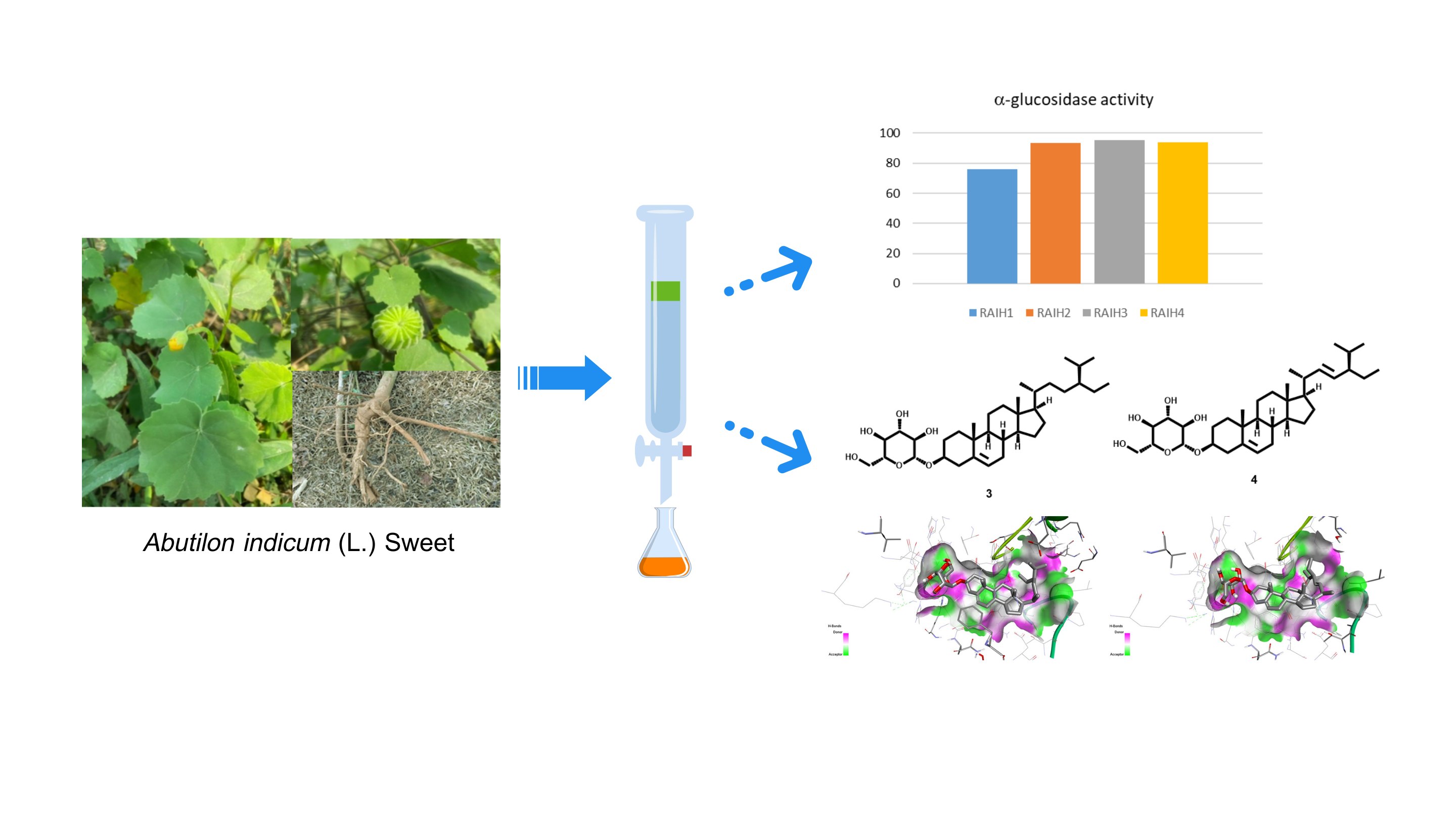
The roots of Abutilon indicum (L.) Sweet were extracted with hexane and subjected to bioassay-guided fractionation to evaluate α-glucosidase inhibitory activity. The hexane extract (RAIH, 0.80% w w-1) showed potent inhibition (88.06%) at 500 mg mL-1. Silica gel chromatography localized the activity to fractions RAIH2–RAIH4 (93.55–95.19% inhibition). Chromatographic separation of the bioactive fractions afforded four known sterol-type constituents, identified by IR, 1H NMR, and 13C NMR, and compared with previously reported data as β-sitosterol (1), stigmasterol (2), daucosterol (3), and stigmasterol-3-O-β-D-glucoside (4). Compounds 3 and 4 were isolated from A. indicum for the first time. Although compounds 1–4 were not assayed for α-glucosidase inhibitory activity in this study, literature data indicate that sterol glycosides are more potent than free sterols. Molecular docking was performed, exhibiting stronger binding for 3 and 4 than for 1 and 2, consistent with a contributory role of sugar moieties. These findings support further compound-level validation. Therefore, investigating this plant and its chemical constituents may enhance the discovery of antidiabetic agents and related therapeutic leads. Collectively, these findings challenge the prevailing focus on polar phenolics as primary α-glucosidase inhibitors and highlight sterol-rich, non-polar fractions as an underexplored yet promising source of antidiabetic leads. This study provides a conceptual framework for expanding enzyme-based antidiabetic screening toward lipophilic phytochemical classes and supports further compound-level validation.
GRAPHICAL ABSTRACT
HIGHLIGHTS
- Bioassay-guided silica gel fractionation localized activity to fractions RAIH2–RAIH4 of 93.55–95.19% inhibition at 500 mg/mL.
- Daucosterol and stigmasterol-3-O-β-D-glucoside were isolated for the first time from Abutilon indicum (L.)
- Molecular docking with yeast α-glucosidase predicted stronger binding for sterol glycosides relative to sterols, supporting the potential role of sugar moieties in enzyme engagement.
References
Toru, H., Mutsunori, F., & Zemin Y. (2019). Postprandial hyperglycemia and postprandial hypertriglyceridemia in type 2 diabetes. Journal of Biomedical Research, 33(1), 1–16. https://doi.org/10.7555/JBR.31.20160164
Dirir, A. M., Daou, M., Yousef, A. F., & Yousef, L. F. (2022). A review of alpha-glucosidase inhibitors from plants as potential candidates for the treatment of type-2 diabetes. Phytochemistry Reviews, 21, 1049–1079. https://doi.org/10.1007/s11101-021-09773-1
Rosak, C., & Mertes, G. (2012) Critical evaluation of the role of acarbose in the treatment of diabetes: patient considerations. Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy, 5, 357–367. https://doi.org/10.2147/DMSO.S28340
Tundis, R., Loizzo, M.R., & Menichini, F. (2010) Natural products as α-amylase and α-glucosidase inhibitors and their hypoglycaemic potential in the treatment of diabetes: an update. Mini-Reviews in Medicinal Chemistry, 10, 315 – 331. https://doi.org/10.2174/138955710791331007
Gamble, J. S. (2011). Flora of the Presidency of Madras. West, Newman and Adlard, https://doi.org/10.5962/bhl.title.21628
Goyal, N., Singh, S., & Sharma, S. (2009). Analgesic effects of various extracts of the root of Abutilon indicum Linn. Journal of Pharmacy and Bioallied Sciences, 1(1), 43. https://doi.org/10.4103/0975-7406.62686
Parekh, J., & Chanda, S. (2007). Antibacterial and phytochemical studies on twelve species of Indian medicinal plants. African Journal of Biomedical Research, 10, 175–181.
Sarkar, R., Haque, A., Ranjan, S., & Sarker, M. (2015). Phytochemical screening, antioxidant and antimicrobial effects of Abutilon indicum (Linn.) leaves extracts. Pharmacologyonline, 1, 94–103.
Rajurkar, R., Jain, R., Matake, N., Aswar, P., & Khadbadi, S. S. (2009). Anti-inflammatory action of Abutilon indicum (L.) Sweet leaves by HRBC membrane stabilization. Research Journal of Pharmacy and Technology, 2(2), 415–416.
Vairavasundaram, R., & Senthil, K. (2009). Antimycotic activity of the components of Abutilon indicum (Malvaceae). Drug Invention Today, 1(2), 137–139.
Ramasubramania, Raja, R., & Kailasam, K. V. (2015). butilon indicum L. (Malvaceae): Medicinal potential review. Pharmacognosy Journal, 7(6), 330–332. https://doi.org/10.5530/pj.2015.6.2
Krisanapun, C., Peungvicha, P., Temsiririrkkul, R., & Wongkrajang, Y. (2009). Aqueous extract of Abutilon indicum Sweet inhibits glucose absorption and stimulates insulin secretion in rodents. Nutrition Research, 29(8), 579–587. https://doi.org/10.1016/j.nutres.2009.07.006
Seetharam, Y., Chalageri, G., & Ramachandra Setty, S. (2002). Hypoglycemic activity of Abutilon indicum leaf extracts in rats. Fitoterapia, 73(2), 156–159. https://doi.org/10.1016/S0367-326X(02)00015-1
Tripathi, P., Chauhan, N. S., & Patel, J. R. (2012). Anti-inflammatory activity of Abutilon indicum extract. Natural Product Research, 26(17), 1659–1661. https://doi.org/10.1080/14786419.2011.616508
Rajput, P., & Patel, K. (2012). Chemical investigation and biological activity of phytoconstituents from methanol extract of Abutilon indicum leaves. Journal of Chemical and Pharmaceutical Research, 4(8), 3959–3965.
Kuo, P. C., Yang, M. L., Wu, P. L., Shih, H. N., Thang, T. D., Dung, N. X., & Wub, T. S. (2008). Chemical constituents from Abutilon indicum. Journal of Asian Natural Products Research, 10(7), 689–693.
Sharma, S. K., & Goyal, N. (2010). Preliminary phytochemical and pharmacognostic profile of Abutilon indicum Linn. root. Der Pharmacia Lettre, 2(5), 308–315.
Mohite, M. S., Shelar, P. A., Raje, V. N., Babar, S. J., & Sapkal, R. K. (2012). Review on pharmacological properties of Abutilon indicum. Asian Journal of Pharmaceutical Research, 2(4), 156–160.
Adhimoolam, K., Sureshbabu, A., Smirnova, E., Muthuramalingam, P., Thi, C. T. D., Senthil, K., & Min, T. (2024). β‐Sitosterol–Dietary sources and role in cancer and diabetes management. Food Science & Nutrition, 12(11), 8870–8886. http://doi:10.1002/fsn3.4380
Durrani, A. K., Khalid, M., Raza, A., Rasool, I. F.U., Khalid, W., Akhtar, M. N., Khan, A. A., Abdullah, Z., & Khadijah, B. (2024). Clinical improvement, toxicity and future prospects of β-sitosterol: a review. CyTA – Journal of Food, 22(1), 2337886. https://doi.org/10.1080/19476337.2024.2337886
Zhang, D., Ge, F., Ji, J., Li, Y. J., Zhang, F. R., Wang, S. Y., Zhang, S. J., Zhang, D. M., & Chen, M. (2023). β-sitosterol alleviates dextran sulfate sodium-induced experimental colitis via inhibition of NLRP3/Caspase-1/GSDMD-mediated pyroptosis. Frontiers in Pharmacology, 14, 1218477. https://doi:10.3389/fphar.2023.1218477
Poulose, N., Sajayan, A., Ravindran, A., Chandran, A., Priyadharshini, G. B., Selvin, J., & Kiran, G. S. (2021). Anti-diabetic potential of a stigmasterol from the seaweed Gelidium spinosum and its application in the formulation of nanoemulsion conjugate for the development of functional biscuits. Frontiers in Nutrition, 16(8), 694362. https://doi: 10.3389/fnut.2021.694362
Li, J., Zheng, X., & Qi, J. (2025). Research progress on the therapeutic mechanisms of stigmasterol for multiple diseases. Molecules, 30(9), 1874. https://doi: 10.3390/molecules30091874
Miya, G. M., Kar, P., Oriola, A. O., & Oyedeji, A. O. (2025). Antidiabetic potential of isolated compounds from Cyperus sexangularis Nees: An in silico molecular docking and dynamic-based approach. Journal of Pharmacy & Pharmacognosy Research, 13(2), 647–661. https://doi.org/10.56499/jppres24.2050_13.2.647
Liang, J., Yang, H., Chen, K., Cai, J., Wang, Y., & Yong, Y. (2025). Therapeutic effect of daucosterol on DSS-induced colitis in mice. BMC Microbiology, 4(26), 18. https://doi: 10.1186/s12866-025-04556-6
Omari, N. E., Jaouadi, I., Lahyaoui, M., Benali, T., Taha, D., Bakrim, S., Menyiy, N. E., Kamari, F. E., Zengin, G., Bangar S. P., Lorenzo, J. M., Gallo, M., Montesano, D., & Bouyahya, A. (2022). Natural sources, pharmacological properties, and health benefits of daucosterol: versatility of actions. Applied Sciences, 12, 5779. https://doi.org/10.3390/app12125779
Mohammed, I. H., Zarial, V., Farimani, M. M., Kharatha, M., Tabefam, M., & Kakey, E. S. I. (2025). In vitro assessment of the antihyperglycemic property of Prosopis farcta J. F. Macbr and Rheum ribes L. and phytochemical profiling of the most active extract. Trends in Phytochemical Research, 9(2), 092511. https://doi.org/10.57647/tpr.2025.0902.11
Pate, A. K., & Kayande, N. R. (2023). An insilico study of stigmasterol glucoside for hypolipidemic activity. Biological Forum – An International Journal, 15(4), 660–667.
Ditchou, Y. O. N., Leutcha, P. B., Miaffo, D., Mamoudou, H., Ali, M. S., Ngnoung, G. A. A., Soh, D., Agrawal, M., Darbawa, R., Tchouboun, E. Z. N., Lannang, A. M., & Noundou, X. S. (2024). In vitro and in silico assessment of antidiabetic and antioxidant potencies of secondary metabolites from Gymnema sylvestre. Biomedicine&Pharmacotherapy, 177, 117043. https://doi.org/10.1016/j.biopha.2024.117043
Zaid, O. A. A., Moawed, F. S. M., Ismail, E. S., & Farrag, M. A. (2023). β-sitosterol attenuates high- fat diet-induced hepatic steatosis in rats by modulating lipid metabolism, inflammation and ER stress pathway. BMC Pharmacology and Toxicology, 24, 31. https://doi.org/10.1186/s40360-023-00671-0
Moradi-Afrapoli, F., Asghari, B., Saeidnia, S., Ajani, Y., Mirjani, M., Malmir, M., Bazaz, R. D., Hadjiakhoondi, A., Salehi, P., Hamburger, M., & Yassa, N. (2012). In vitro α-glucosidase inhibitory activity of phenolic constituents from aerial parts of Polygonum hyrcanicum. DARU Journal of Pharmaceutical Sciences, 20, 37. https://doi.org/10.1186/2008-2231-20-37
Yamamoto, K., Miyake, H., Kusunoki, M., & Osaki, S. (2010). Crystal structures of isomaltase from Saccharomyces cerevisiae and in complex with its competitive inhibitor maltose. FEBS Journal, 277, 4205–4214.
Weiner, S. J., Kollman, P. A., Case, D. A., Singh, U. C., Ghio, C., Alahona, G., Profeta, S., & Weiner, P. (1984). A new force field for molecular mechanical simulation of nucleic acids and proteins. Journal of the American Chemical Society, 106, 765–784.
Morris, G. M., Goodsell, D. S., Halliday, R. S., Huey, R., Hart, W. E., Belew, R. K., & Olson, A. J. (1998). Automated docking using a lamarckian genetic algorithm and an empirical binding free energy function. Journal of Computational Chemistry, 19, 1639–1662.
Yamamoto, K., Miyake, H., Kusunoki, M., & Osaki, S. (2010). Crystal structures of isomaltase from Saccharomyces cerevisiae and in complex with its competitive inhibitor maltose. FEBS Journal, 277, 4205–4214.
Chaturvedula, V. S. P., & Prakash, I. (2012). Isolation of Stigmasterol and B -sitosterol from the dichlomethane extract of Rubus suavissmus. International Current Pharmaceutical Journal, 1(9):239–242.
Esmaeili, M. A., & Farimani, M. M. (2014). Inactivation of PI3K/Akt pathway and upregulation of PTEN gene are involved in daucosterol-induced apoptosis in human breast adenocarcinoma cells. South African Journal of Botany, 93, 37–47. https://doi.org/10.1016/j.sajb.2014.03.010
Afrizal, S., & Mai, E. (2015). Isolation and elucidation structure of stigmasterol glycoside from Nothopanax scutellarium Merr leaves. Journal of Chemical and Pharmaceutical Research, 7(12), 763–765.
Tabussum, A., Riaz, N., Saleem, M., Ashraf, M., Ahmad, M., Alam, U., Jabeen, B., Malik, A., & Jabbar, A. (2013). α-Glucosidase inhibitory constituents from Chrozophora plicata. Phytochemistry Letters, 6(4), 614–619 http://doi.org/10.1016/j.phytol.2013.08.005
Murugesu, S., Ibrahim, Z., Ahmed, Q. U., Yusoff, N. I. N., Uzir B. F., Perumal, V., Abas, F., Saari, K., Hesham, E. S., & Khatib, A. (2018). Characterization of -glucosidase inhibitors from Clinacanthus nutans Lindau leaves by Gas Chromatography-Mass Spectrometry-Based metabolomics and molecular docking simulation. Molecules, 23, 2402. http://doi.org/10.3390/molecules23092402
Gu, Y., Yang, X., Shang, C., Thao, T. T. P., & Koyama, T. (2021). Inhibitory properties of saponin from Eleocharis dulcis peel against α-glucosidase. RSC Advances, 11, 15400–15409. http://doi.org/10.1039/d1ra02198b

Downloads
Additional Files
Published
How to Cite
Issue
Section
Categories
License
Copyright (c) 2026 Creative Science

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.










