Reduction of Cr(VI) with Infrared Light and Chemical Adsorption of Cr(III) by a God-Crown/Bentonite Composite for Electroplating Waste Remediation
Main Article Content
Abstract
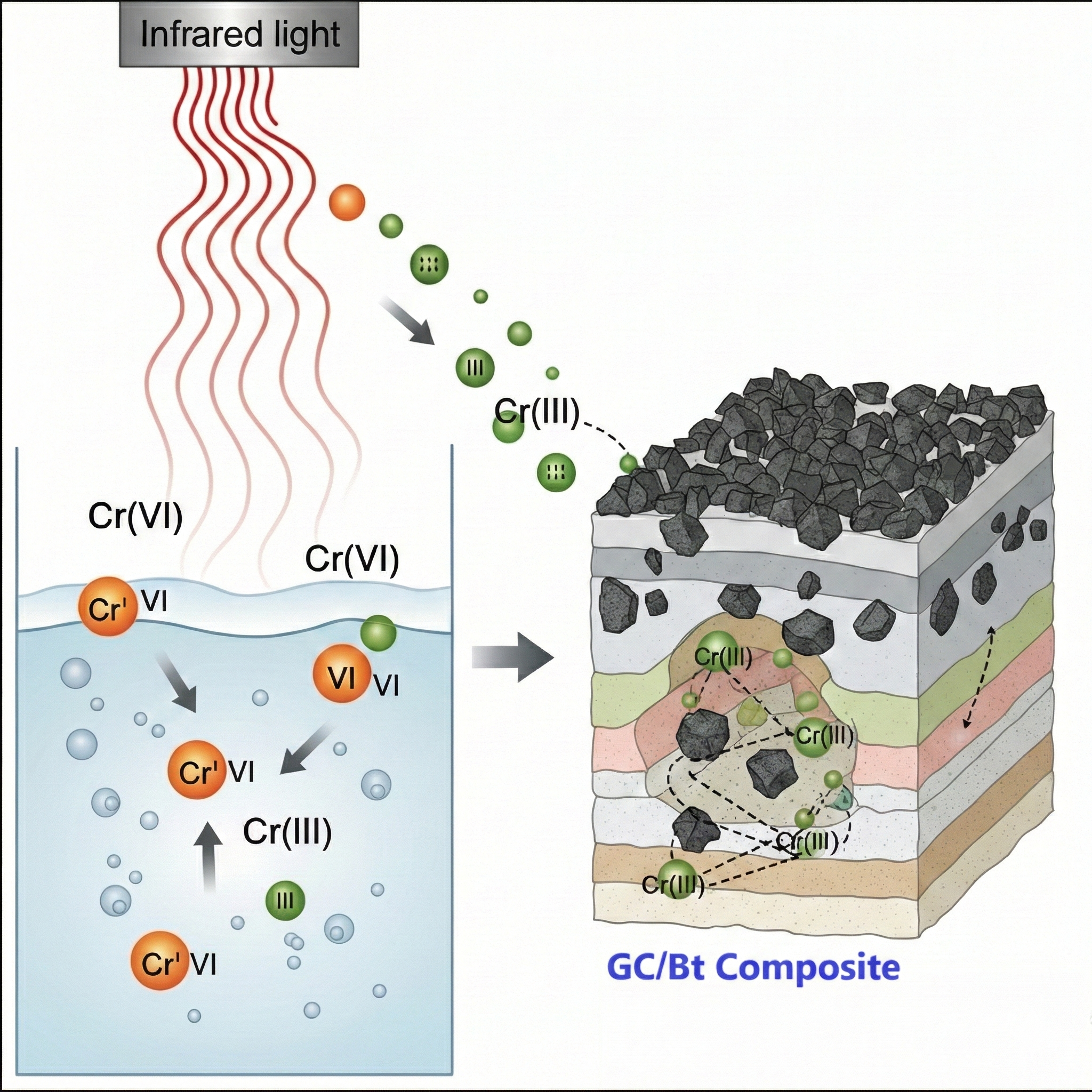
Heavy metal pollution, particularly chromium (Cr) from electroplating industrial waste, has severely threatened environmental quality and human health. This study aims to develop a composite adsorbent material based on bentonite and god crown biomass capable of removing chromium ions from liquid waste through a combination of reduction and adsorption mechanisms. The god crown/bentonite (GC/Bt) composite was synthesized at a mass ratio of 2:1 and calcined at 900°C. FTIR characterization revealed active functional groups (–OH, C=O, Si–O, and Al–O–Si), whereas BET analysis revealed a mesoporous structure (surface area 31.12 m2 g-1, pore diameter 4.37 nm) suitable for ion diffusion. The reduction of Cr(VI) to Cr(III) was facilitated by infrared (IR) irradiation, with 950 nm identified as the optimum wavelength compared with 730 nm. This conversion was confirmed by the significant increase in removal efficiency, as Cr(III) is more readily adsorbed by the composite than Cr(VI). The integrated system achieved a maximum removal efficiency (%R) of 86% and an adsorption capacity (qe) of 703.19 mg g-1 under optimal continuous column conditions (bed height of 30 cm and flow rate of 4 L min-1). The isotherm study showed the best fit with the Freundlich model, indicating a heterogeneous adsorbent surface, whereas the adsorption kinetics followed the pseudo-second-order model (R2>0.97), indicating the dominance of the chemisorption mechanism. These results confirm that combining infrared reduction and chemical adsorption by GC/Bt composites is a practical approach for industrial chromium waste treatment.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Published articles are under the copyright of the Applied Environmental Research effective when the article is accepted for publication thus granting Applied Environmental Research all rights for the work so that both parties may be protected from the consequences of unauthorized use. Partially or totally publication of an article elsewhere is possible only after the consent from the editors.
References
Hanui Yang, Namgyu Kim, and Donghee Park, “Superior Removal of Toxic Cr(VI) from Wastewaters by Natural Pine Bark,” Separations, vol. 10, no. 8, pp. 430, July 2023, doi: 10.3390/separations10080430.
Xuan Phuong Nguyen, Dinh Tuyen Nguyen, Van Viet Pham, and Viet Duc Bui, “EVALUATION OF THE SYNERGISTIC EFFECT IN WASTEWATER TREATMENT FROM SHIPS BY THE ADVANCED COMBINATION SYSTEM,” Water Conservation & Management, vol. 5, no. 1, pp. 60–65, 2021, doi: 10.26480/wcm.01.2021.60.65.
N.K. Lazaridis, D.N. Bakoyannakis, and E.A. Deliyanni, “Chromium(VI) sorptive removal from aqueous solutions by nanocrystalline akaganèite,” Chemosphere, vol. 58, no. 1, pp. 65–73, January 2005, doi: 10.1016/j.chemosphere.2004.09.007.
Marjorie Salvador De Luna, Ian Auza Navarrete, Adonis Pasia Adornado, Lemmuel Lara Tayo, Allan Nana Soriano, and Rugi Vicente Del Castillo Rubi, “Influence of Nutrient Supplement in the Single Heavy Metal (Pb2+, Cd2+, Cr3+) Uptake and Mineral Nutrients Absorption by Water Kangkong (Ipomoea aquatica forsk),” Applied Science and Engineering Progress, June 2022, doi: 10.14416/j.asep.2022.06.001.
R. Senthilkumar, K. Vijayaraghavan, J. Jegan, and M. Velan, “Batch and column removal of total chromium from aqueous solution using Sargassum polycystum,” Environmental Progress & Sustainable Energy, vol. 29, no. 3, pp. 334–341, October 2010, doi: 10.1002/ep.10416.
M. K. Rai, B. S. Giri, Y. Nath, H. Bajaj, S. Soni, R. P. Singh, R. S. Singh, and B. N. Rai, “Adsorption of hexavalent chromium from aqueous solution by activated carbon prepared from almond shell: kinetics, equilibrium and thermodynamics study,” Journal of Water Supply: Research and Technology-Aqua, vol. 67, no. 8, pp. 724–737, December 2018, doi: 10.2166/aqua.2018.047.
Tamirat Dula, Khalid Siraj, and Shimeles Addisu Kitte, “Adsorption of Hexavalent Chromium from Aqueous Solution Using Chemically Activated Carbon Prepared from Locally Available Waste of Bamboo ( Oxytenanthera abyssinica ),” ISRN Environmental Chemistry, vol. 2014, pp. 1–9, February 2014, doi: 10.1155/2014/438245.
T. P. Dhungana, and P. N. Yadav, “Determination of Chromium in Tannery Effluent and Study of Adsorption of Cr (VI) on Saw dust and Charcoal from Sugarcane Bagasses,” Journal of Nepal Chemical Society, vol. 23, pp. 93–101, January 1970, doi: 10.3126/jncs.v23i0.2102.
Muhammad Imran, Zia Ul Haq Khan, Muhammad Mohsin Iqbal, Jibran Iqbal, Noor Samad Shah, Saba Munawar, Shafaqat Ali, Behzad Murtaza, Muhammad Asif Naeem, and Muhammad Rizwan, “Effect of biochar modified with magnetite nanoparticles and HNO3 for efficient removal of Cr(VI) from contaminated water: A batch and column scale study,” Environmental Pollution, vol. 261, no. NA, pp. 114231, 2020, doi: 10.1016/j.envpol.2020.114231.
Jianyang Song, Qiulai He, Xiaoling Hu, Wei Zhang, Chunyan Wang, Rongfan Chen, Hongyu Wang, and Ahmed Mosa, “Highly efficient removal of cr(VI) and cu(II) by biochar derived from artemisia argyi stem,” Environmental Science and Pollution Research, vol. 26, no. 13, pp. 13221–13234, 2019, doi: 10.1007/s11356-019-04863-2.
Jingchun Tang, Beibei Zhao, Honghong Lyu, and Ding Li, “Development of a novel pyrite/biochar composite (BM-FeS2@BC) by ball milling for aqueous Cr(VI) removal and its mechanisms,” Journal of Hazardous Materials, vol. 413, no. NA, pp. 125415-NA, 2021, doi: 10.1016/j.jhazmat.2021.125415.
Danlian Huang, Caihong Liu, Chen Zhang, Rui Deng, Rongzhong Wang, Wenjing Xue, Hao Luo, Guangming Zeng, Qing Zhang, and Xueying Guo, “Cr(VI) removal from aqueous solution using biochar modified with Mg/Al-layered double hydroxide intercalated with ethylenediaminetetraacetic acid,” Bioresource Technology, vol. 276, no. NA, pp. 127–132, 2019, doi: 10.1016/j.biortech.2018.12.114.
Tolera Seda Badessa, Esayas Wakuma, and Ali Mohammed Yimer, “Bio-sorption for effective removal of chromium(VI) from wastewater using Moringa stenopetala seed powder (MSSP) and banana peel powder (BPP),” BMC Chemistry, vol. 14, no. 1, pp. 71, December 2020, doi: 10.1186/s13065-020-00724-z.
Prashanta Talukder, Razia Sultana, Mustafizur Rahman Naim, Plabon Islam Turzo, and Umme Habiba Bodrun Naher, “Optimization of batch process parameters for chromium (VI) removal from synthetic wastewater using eggshell–clay composite,” Discover Applied Sciences, vol. 6, no. 6, pp. 291, May 2024, doi: 10.1007/s42452-024-05865-7.
Shivshankar R. Kondagorla, and Swapnil V. Madhamshettiwar, “Adsorption of Chromium (VI) from Wastewater Using Blended Natural Adsorbent,” Oriental Journal Of Chemistry, vol. 40, no. 4, pp. 1134–1137, August 2024, doi: 10.13005/ojc/400427.
Senthil Rajan, Ramesh Kumar, S Vivek, Gokulan R, and Balaji G, “Biosorption of chromium (VI+) using tamarind fruit shells in continuously mixed batch reactor,” Global NEST Journal, vol. 25, no. 1, pp. 180–186, November 2022, doi: 10.30955/gnj.004561.
Muhammad Ilyas, Waqas Ahmad, Hizbullah Khan, and Saeeda Yousaf, “Potentially poisonous elements removal from vehicle-wash wastewater and aqueous solutions using composite adsorbents,” Desalination and Water Treatment, vol. 224, no. NA, pp. 331–342, 2021, doi: 10.5004/dwt.2021.27179.
Mohammad Ali Rezvani, Parisa Afshari, and Masomeh Aghmasheh, “Deep catalytic oxidative desulfurization process catalyzed by TBA-PWFe@NiO@BNT composite material as an efficient and recyclable phase-transfer nanocatalyst,” Materials Chemistry and Physics, vol. 267, pp. 124662, July 2021, doi: 10.1016/j.matchemphys.2021.124662.
My Slimane Slimani, Mustapha Aazza, Hamid Barkouch, Meryem Amar, Abdelaaziz Alioui, Ouafa Tahiri Alaoui, Si Mohamed Bouzzine, and Hammou Ahlafi, “Bentonite and organobentonite nanocomposite for removing chromium species from aqueous solutions,” Applied Clay Science, vol. 267, pp. 107736, April 2025, doi: 10.1016/j.clay.2025.107736.
Radheshyam R. Pawar, Prabuddha Gupta, Lalhmunsiama, Hari C. Bajaj, and Seung Mok Lee, “Al-intercalated acid activated bentonite beads for the removal of aqueous phosphate,” Science of the Total Environment, vol. 572, pp. 1222–1230, 2016, doi: 10.1016/j.scitotenv.2016.08.040.
Ali Napiah Nasution, Yasherly Amrina, Rahmiana Zein, Hermansyah Aziz, and Edison Munaf, “Biosorption characteristics of Cd(II) ions using herbal plant of mahkota dewa (Phaleria macrocarpa),” Journal of Chemical and Pharmaceutical Research, vol. 7, no. 7, pp. 189–196, 2015
Rosliza Ahmad, Mohd Khairul Nizam Mazlan, Amir Firdaus Abdul Aziz, Amirah Mohd Gazzali, Mira Syahfriena Amir Rawa, and Habibah A. Wahab, “Phaleria macrocarpa (Scheff.) Boerl.: An updated review of pharmacological effects, toxicity studies, and separation techniques,” Saudi Pharmaceutical Journal, vol. 31, no. 6, pp. 874–888, June 2023, doi: 10.1016/j.jsps.2023.04.006.
Neway Belachew, and Hirpo Hinsene, “Preparation of cationic surfactant-modified kaolin for enhanced adsorption of hexavalent chromium from aqueous solution,” Applied Water Science, vol. 10, no. 1, pp. 38, January 2020, doi: 10.1007/s13201-019-1121-7.
Nabil Bougdah, Nabil Messikh, Salim Bousba, Fayçal Djazi, Pierre Magri, and Marek Rogalski, “Removal of chlorobenzene by adsorption from aqueous solutions on the HDTMA-bentonites as a function of HDTMA/CEC ratio,” Current Research in Green and Sustainable Chemistry, vol. 3, pp. 100038, June 2020, doi: 10.1016/j.crgsc.2020.100038.
Friedrich Fink, Franziska Emmerling, and Jana Falkenhagen, “Identification and Classification of Technical Lignins by means of Principle Component Analysis and k‐Nearest Neighbor Algorithm,” Chemistry–Methods, vol. 1, no. 8, pp. 354–361, August 2021, doi: 10.1002/cmtd.202100028.
S. G. Kostryukov, H. B. Matyakubov, Yu. Yu. Masterova, A. Sh. Kozlov, M. K. Pryanichnikova, A. A. Pynenkov, and N. A. Khluchina, “Determination of Lignin, Cellulose, and Hemicellulose in Plant Materials by FTIR Spectroscopy,” Journal of Analytical Chemistry, vol. 78, no. 6, pp. 718–727, June 2023, doi: 10.1134/S1061934823040093.
A. Tabak, N. Yilmaz, E. Eren, B. Caglar, B. Afsin, and A. Sarihan, “Structural analysis of naproxen-intercalated bentonite (Unye),” Chemical Engineering Journal, vol. 174, no. 1, pp. 281–288, October 2011, doi: 10.1016/j.cej.2011.09.027.
Ihssane Belbachir, and Benamar Makhoukhi, “Adsorption of Bezathren dyes onto sodic bentonite from aqueous solutions,” Journal of the Taiwan Institute of Chemical Engineers, vol. 75, pp. 105–111, June 2017, doi: 10.1016/j.jtice.2016.09.042.
Dagmawi Mulugeta Degefu, and Mekibib Dawit, “Chromium Removal from Modjo Tannery Wastewater Using Moringa stenopetala Seed Powder as an Adsorbent,” Water, Air, & Soil Pollution, vol. 224, no. 12, pp. 1719, December 2013, doi: 10.1007/s11270-013-1719-6.
Pratima Meshram, Sushanta Kumar Sahu, Banshi Dhar Pandey, Vinay Kumar, and Tilak Raj Mankhand, “Removal of Chromium(III) from the Waste Solution of an Indian Tannery by Amberlite IR 120 Resin,” International Journal of Nonferrous Metallurgy, vol. 01, no. 03, pp. 32–41, 2012, doi: 10.4236/ijnm.2012.13005.
Weikang Liu, Liang Yang, Shihao Xu, Yao Chen, Bianhua Liu, Zhong Li, and Changlong Jiang, “Efficient removal of hexavalent chromium from water by an adsorption–reduction mechanism with sandwiched nanocomposites,” RSC Advances, vol. 8, no. 27, pp. 15087–15093, 2018, doi: 10.1039/C8RA01805G.