Activated Carbon Prepared from Residual Eucalyptus Wood for the Removal of Methyl Orange: Equilibrium, Kinetics, Thermodynamics, and Desorption
Main Article Content
Abstract
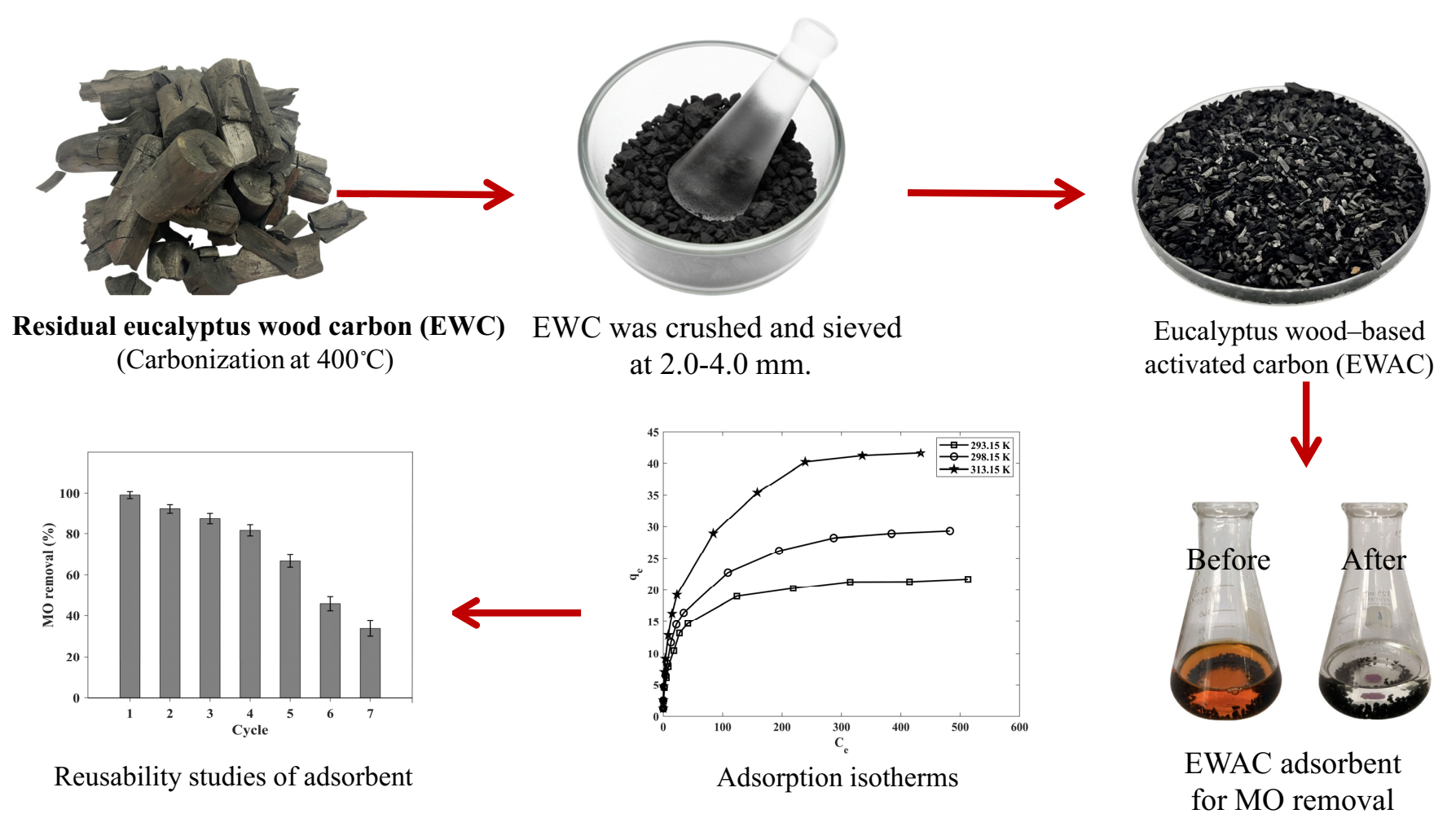
This research utilizes activated carbon derived from residual Eucalyptus wood (EW) for the removal of methyl orange (MO). The residual Eucalyptus wood-based activated carbon (EWAC) was produced through carbonization at 400 °C (EWC) followed by activation with a mass ratio of H3PO4 to EWC of 1:3, which was conducted at 800 °C for 1 h. The characterization of the EWAC involved various analytical techniques, including FTIR, XRD, BET, and CHN analysis. The adsorption parameters, such as pH (range: 3–8), adsorbent dose (range: 0.01–0.5 g per 50 mL of MO solution), contact time (range: 5–720 min), initial MO concentration (range: 5–600 mg L-1), and temperature (range: 20–40 °C), were investigated. The kinetic study suggested that the adsorption behavior correlated well with the pseudo-second-order model, indicating a suitable fit for the experimental data. The intraparticle diffusion analysis suggested that the external film diffusion of dye molecules controls the overall adsorption rate. Moreover, the adsorption model provided good fit with the Langmuir isotherm model, confirming monolayer adsorption with an adsorption capacity (qmax) of 29.11 mg g-1 at 303.15 K. Thermodynamic studies also confirmed that endothermic and chemisorption processes are favored for the adsorption process even at high temperatures. Furthermore, the EWAC demonstrated potential for regeneration and reuse over four operational cycles, highlighting its cost-effectiveness and eco-friendly nature for removing MO from synthetic dyes in wastewater.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Published articles are under the copyright of the Applied Environmental Research effective when the article is accepted for publication thus granting Applied Environmental Research all rights for the work so that both parties may be protected from the consequences of unauthorized use. Partially or totally publication of an article elsewhere is possible only after the consent from the editors.