Coating Ag2S Films on Silver Jewelry by Electrochemical Method
DOI:
https://doi.org/10.55674/ias.v10i2.242673Keywords:
Jewelry, Silver, Electrochemical, Coating, Ag2SAbstract
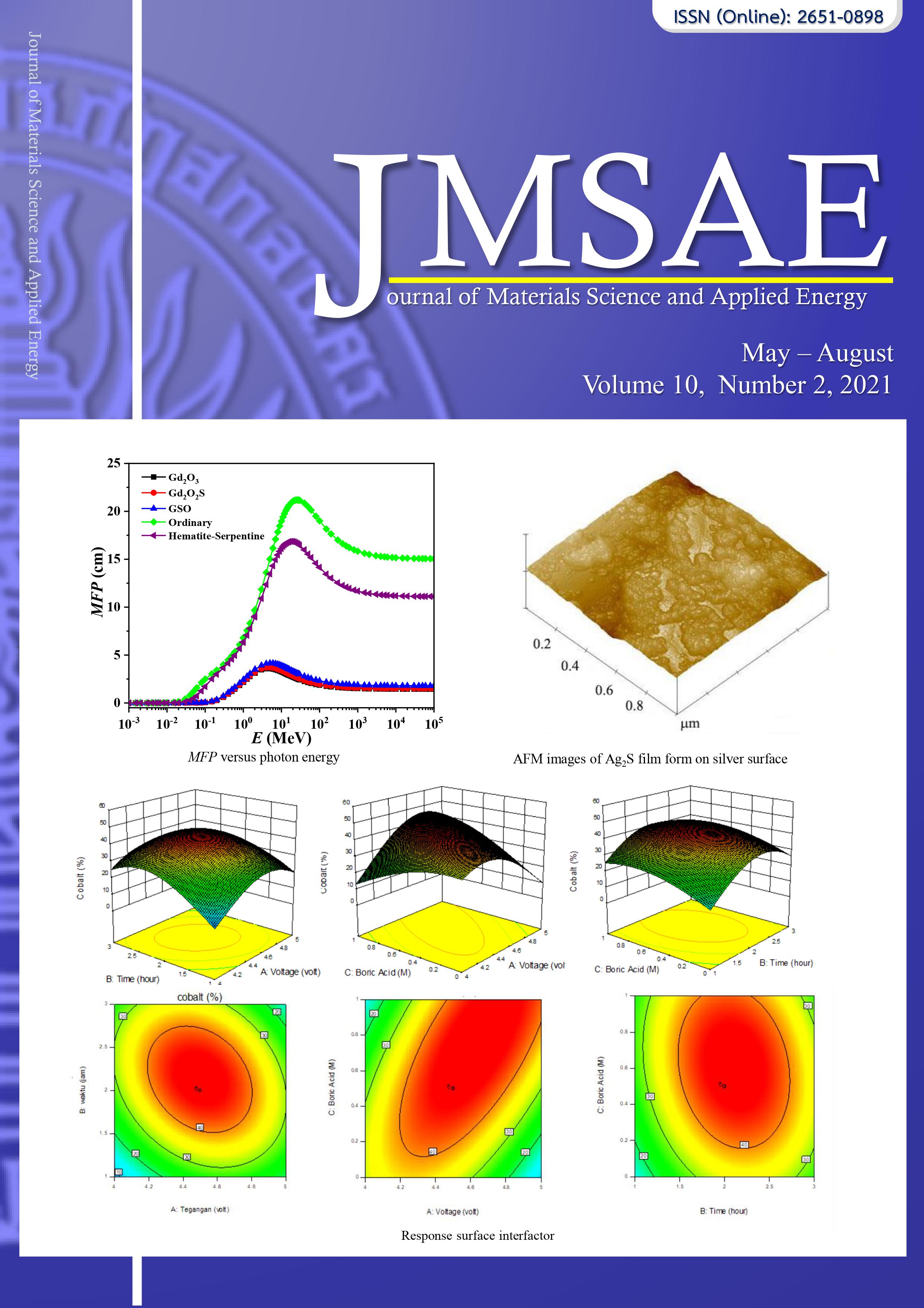
Ag2S films were fabricated on the silver surface by electrochemical deposition from the mixture electrolyte solution including Sulfur and Sodium Hydroxide at room temperature. The composition and surface morphology of the Ag2S films were investigated with various voltage from 1.50 V and 2 V for 30 – 150 s. The SEM images show that Ag2S films are uniform with high compact at 1.50 V for 150 s. Moreover, the density of film tends to increase as the deposition time of immersion in electrolyte solution increases, and applied voltage up to 2 V. The AFM images show that the grain size and root mean square (RMS) roughness decrease with the increase of voltage value and deposition time of immersion. The CIE Lab results were showed that the color of Ag2S films could be changed color from red-blue to dark-yellow-green. The applied voltage and deposition time are 1.50 V at 150 s, respectively for depositing Ag2S films on the silver jewelry products.